12.6 Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
(i) PhMgBr and then H3O+
(ii) Tollens’ reagent
(iii) Semicarbazide and weak acid
(iv) Excess ethanol and acid
(V) Zinc amalgam and dilute hydrochloric acid
The structure of cyclohexanecarbaldehyde is as follows:
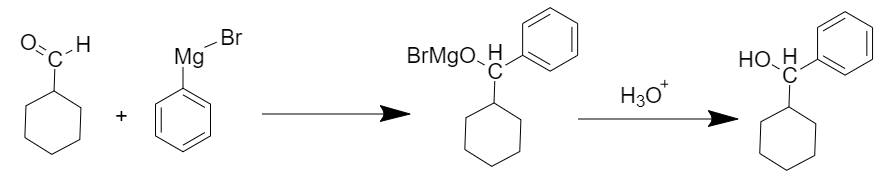
(i)
When cyclohexanecarbaldehyde reacts with the Grignard reagent followed by acidic hydrolysis then secondary alcohol is obtained as a product.
The reaction is as follows:
Cyclohexylphenylcarbinol
(ii)
The tollens' reagent is an ammoniacal silver nitrate solution. When cyclohexanecarbaldehyde reacts with tollens' reagent then carboxylate ion with silver mirror is obtained as a product.
The reaction is as follows:
(iii)
When cyclohexanecarbaldehyde reacts with semicarbazide in the acidic medium then semicarbazone is obtained as a product.
The reaction is as follows:
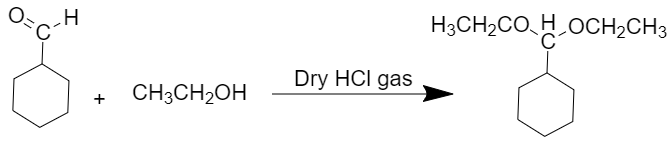
(iv)
When cyclohexanecarbaldehyde reacts with an excess of ethanol in the presence of dry gas then acetal is obtained as a product.
The reaction is as follows:
(v)
When cyclohexanecarbaldehyde undergoes

© 2026 GoodEd Technologies Pvt. Ltd.