Match the item in column I and in column II.
Column I
Column II
a.
No heat is absorbed by the system from the surroundings, but work (w) is done on the system.
i.
∆U = q – w, closed system.
b.
No work is done on the system, but q amount of heat is taken out from the system and given to the surroundings.
ii.
∆U = wad, a wall is adiabatic.
c.
w amount of work is done by the system and q amount of heat is supplied to the system.
iii.
∆U = –q, thermally conducting walls.
1.
a = i; b = ii; c = iii
2.
a = ii; b = i; c = iii
3.
a = ii; b = iii; c = i
4.
a = iii; b = ii; c = i
| (i) | When liquid crystallizes into a solid, entropy increases. |
| (ii) | When the temperature of a crystalline solid is raised from 0 K to 115 K then entropy increases. |
| (iii) | 2 NaHCO3 (s) →Na2CO3 (s) +CO2(g)+H2O(g); Entropy increases. |
| (iv) | H2(g)→2H(g) ; Entropy decreases. |
For one mole of an ideal gas, which of these statements must be true?
(I) U and H each depend only on temperature.
(II) Compressibility factor z is not equal to 1.
(III) CP, m – CV, m = R
(IV) dU = CVdT for any process.
1. (I), (III) and (IV)
2. (II), (III) and (IV)
3. (III) and (IV)
4. (I) and (III)
1.0 mol of a monoatomic ideal gas is expanded from state (1) to state (2) as shown in the graph below:
The work done for the expansion of gas from state (1) to state (2) at 298 K will be
1. 1617.6 J
2. -1617.6 J
3. 1717.6 J
4. -1717.6 J
An enthalpy diagram for a particular reaction is given below:
The correct statement among the following is-
1. Reaction is spontaneous
2. Reaction is non-spontaneous
3. Cannot predict spontaneity of the reaction from the graph given above
4. None of the above
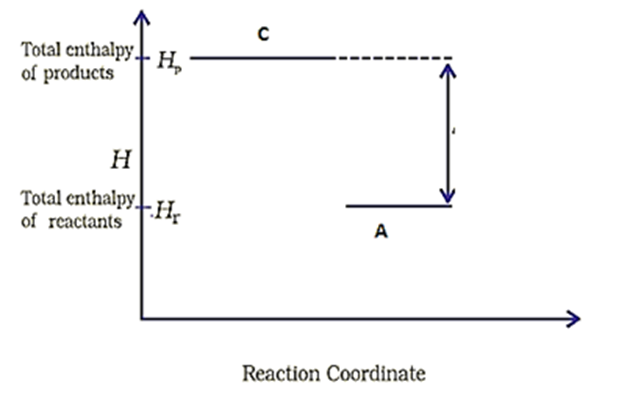
Consider the following diagram for a reaction
The nature of the reaction is-
| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of the above |
Consider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above
Consider the following graph.
The work done shown by the above-mentioned graph is-
| 1. | Positive | 2. | Negative |
| 3. | Zero | 4. | Cannot be determined |
For the graph given below, it can be concluded that work done during the process shown will be-
| 1. | Zero | 2. | Negative |
| 3. | Positive | 4. | Cannot be determined |
The correct statements among the following are :
| a. | The enthalpy of two moles of ZnO is less than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ |
| b. | The enthalpy of two moles of ZnO is more than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ |
| c. | 693 . 8 kJ mol -1 energy is evolved in the reaction |
| d. | 693 . 8 kJ mol -1 energy is absorbed in the reaction |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)