From a certain apparatus, the diffusion rate of hydrogen has an average value of 28.7 . The diffusion of another gas under the same conditions is measured to have an average rate of 7.2 . The unknown gas is:
1. Oxygen
2. Nitrogen
3. Helium
4. None of these
1. Oxygen
2. Nitrogen
3. Helium
4. None of these
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at \(-20^\circ \mathrm{C}?\)
(Given the atomic mass of \(\text{Ar}=39.9~\text{u}\) and of \(\text{He}=4.0~\text{u}\)).
1. \(1.01 \times 10^3 ~\text{K} \)
2. \(3.15 \times 10^3 ~\text{K} \)
3. \(1.91 \times 10^3~ \text{K} \)
4. \(2.52 \times 10^3 ~\mathrm{K}\)
What is the average thermal energy of a helium atom at room temperature (\(27^{\circ}\mathrm{C}\))?
| 1. | \(11 . 21 \times 10^{- 20} \text{J}\) | 2. | \(3 . 09 \times 10^{- 16} \text{J}\) |
| 3. | \( 6 . 21 \times 10^{- 21} \text{J} \) | 4. | \(5 . 97 \times 10^{- 19} \text{J}\) |
What is the total number of air molecules (inclusive of oxygen, nitrogen, water vapor, and other constituents) in a room of capacity \(25.0\) m3 at a temperature of \(27^\circ \mathrm C\) and \(1\) atm pressure?
1. \(6.1\times10^{23}\) molecules
2. \(6.1\times10^{26}\) molecules
3. \(7.1\times10^{23}\) molecules
4. \(7.1\times10^{26}\) molecules
An air bubble of volume 1.0 rises from the bottom of a lake 40 m deep at a temperature of 12 °C. To what volume does it grow when it reaches the surface, which is at a temperature of 35 °C?
1. 5.3 cm3
2. 4.0 cm3
3. 3.7 cm3
4. 4.9 cm3
An oxygen cylinder of volume 30 litres has an initial gauge pressure of 15 atm and a temperature of 27 °C. After some oxygen is withdrawn from the cylinder, the gauge pressure drops to 11 atm, and its temperature drops to 17 °C. The mass of oxygen taken out of the cylinder is:
1. 0.14 kg
2. 0.16 kg
3. 0.18 kg
4. 0.21 kg
The figure shows a plot of PV/T versus P for of oxygen gas at two different temperatures.
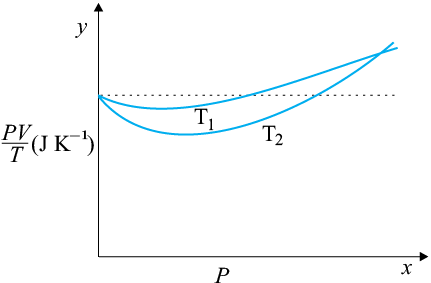
The value of PV/T where the curves meet on the y-axis is:
The figure shows a plot of \(\dfrac{PV}{T}\) versus \(P\) for \(1.00\times10^{-3} \) kg of oxygen gas at two different temperatures.
Then relation between \(T_1\) and \(T_2\) is:
1. \(T_1=T_2\)
2. \(T_1<T_2\)
3. \(T_1>T_2\)
4. \(T_1 \geq T_2\)
Molar volume is the volume occupied by \(1\) mol of any (ideal) gas at standard temperature and pressure is:
(STP: \(1\) atmospheric pressure, \(0~^\circ \text{C}\))
1. \(0\)
2. \(22.4\) litres
3. \(11.2\) litres
4. \(1\) litres
The fraction of molecular volume to the actual volume occupied by oxygen gas at STP is: (Take the diameter of an oxygen molecule to be 3 Å).
1. 4 × 10−4
2. 5 × 10−4
3. 3 × 10−4
4. 1 × 10−4



