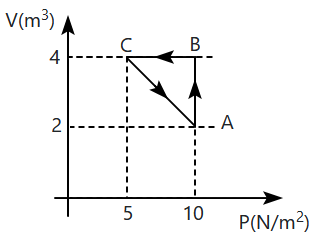
An ideal gas undergoes a cyclic process ABCA as shown. The heat exchange between the system and the surrounding during the process will be:
| 1. | 10 J | 2. | 5 J |
| 3. | 15 J | 4. | 20 J |
A thermodynamic system undergoes a cyclic process \(ABCDA\) as shown in Fig. The work done by the system in the cycle is:
1. \( P_0 V_0 \)
2. \( 2 P_0 V_0 \)
3. \( \frac{P_0 V_0}{2} \)
4. zero
\(ABCA\) is a cyclic process. Its \(P\text-V\) graph would be:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
In the cyclic process shown in the pressure-volume \((P-V)\) diagram, the change in internal energy is equal to:
1.
2.
3.
4. zero
Which one of the following is correct for one complete cycle of a thermodynamic process on a gas as shown in the \((P-V)\) diagram?
| 1. | \(\Delta E_{int}= 0, Q<0\) | 2. | \(\Delta E_{int}= 0, Q>0\) |
| 3. | \(\Delta E_{int}>0, Q<0\) | 4. | \(\Delta E_{int}< 0, Q>0\) |
An ideal gas is taken through the cycle as shown in figure below. If the net heat supplied to the gas is 10 J, then the work done by the gas in the process is
| 1. | -10 J | 2. | -30 J |
| 3. | -15 J | 4. | -20 J |
For the indicator diagram given below, which of the following is not correct?
| 1. | Cycle - II is a heat engine cycle. |
| 2. | Net work is done on the gas in cycle I. |
| 3. | Work done is positive for cycle I. |
| 4. | Work done is positive for cycle II. |
In the P-V diagram shown, the gas does 5 J of work in the isothermal process ab and 4 J in the adiabatic process bc. What will be the change in internal energy of the gas in the straight path from c to a?
1. 9J
2. 1 J
3. 4 J
4. 5 J
In a cyclic process, the internal energy of the gas:
| 1. | Increases | 2. | Decreases |
| 3. | Remains constant | 4. | Becomes zero |
If in the thermodynamic process shown in the figure, the work done by the system along A B C is 50 J and the change in internal energy during C A is 30 J, then the heat supplied during A B C is:
| 1. | 50 J | 2. | 20 J |
| 3. | 10 J | 4. | 80 J |