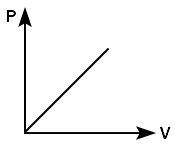
Which of the following graph shows the variation of pressure P with volume V for an ideal gas at a constant temperature?
1.

2.

3.

4.





To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
A gas performs the minimum work when it expands:
| 1. | Isochorically | 2. | Isobarically |
| 3. | Adiabatically | 4. | Isothermally |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
An ideal gas goes from A to B via two processes, l and ll, as shown. If and are the changes in internal energies in processes I and II, respectively, then (\(P:\) pressure, \(V:\) volume)
| 1. | ∆U1 > ∆U2 | 2. | ∆U1 < ∆U2 |
| 3. | ∆U1 = ∆U2 | 4. | ∆U1 ≤ ∆U2 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
An ideal monoatomic gas \(\left(\gamma = \frac{5}{3}\right )\) absorbs 50 cal in an isochoric process. The increase in internal energy of the gas is:
| 1. | 20 cal | 2. | Zero |
| 3. | 50 cal | 4. | 30 cal |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The pressure-temperature (P-T) graph for two processes, A and B, in a system is shown in the figure. If and are work done by the gas in process A and B respectively, then:
| 1. | \(W_{1}\) = \(W_{2}\) | 2. | \(W_{1}\) < \(W_{2}\) |
| 3. | \(W_{1}\) > \(W_{2}\) | 4. | \(W_{1}\) = \(-W_{2}\) |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The variation of molar heat capacity at constant volume with temperature T for a monatomic gas is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
When a system is moved from state a to state b along the path acb, it is discovered that the system absorbs 200 J of heat and performs 80 J of work. Along the path adb, heat absorbed Q = 144 J. The work done along the path adb is:
| 1. | 6 J | 2. | 12 J |
| 3. | 18 J | 4. | 24 J |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
If a refrigerator extracts heat 'a' from the cold reservoir and 'b' is the heat released from the hot reservoir, then the work done on the refrigerant (system) is:
1. a + b
2.
3. a
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Heat is supplied to a diatomic gas in an isochoric process. The ratio is: (symbols have usual meanings)
1. 5 : 3
2. 5: 2
3. 1: 1
4. 5: 7

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
In the cyclic process shown in the pressure-volume \((P-V)\) diagram, the change in internal energy is equal to:
1.
2.
3.
4. zero

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.






