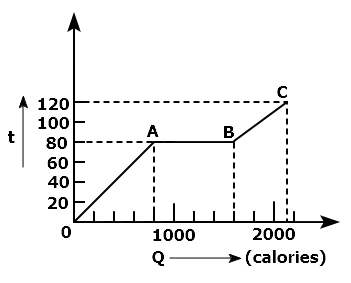
A substance is in solid form at \(0^{\circ}\mathrm{C}\). The amount of heat added to this substance and its temperature are plotted in the following graph. If the relative specific heat capacity of the solid substance is 0.5, from the graph, the specific latent heat of the melting process is: (Specific heat capacity of water = 1000 cal kg-1 K-1 )
1.
60000 cal kg-1
2.
40000 cal kg-1
3.
10000 cal kg-1
4.
20000 cal kg-1

A copper rod of \(88\) cm and an aluminium rod of an unknown length have an equal increase in their lengths independent of an increase in temperature. The length of the aluminium rod is:
\(\left(\alpha_{Cu}= 1.7\times10^{-5}~\text{K}^{-1}~\text{and}~\alpha_{Al}= 2.2\times10^{-5}~\text{K}^{-1}\right)\)
1. \(68~\text{cm}\)
2. \(6.8~\text{cm}\)
3. \(113.9~\text{cm}\)
4. \(88~\text{cm}\)
On a new scale of temperature, which is linear and called the \(\mathrm{W}\) scale, the freezing and boiling points of water are \(39^\circ ~\mathrm{W}\)and \(239^\circ ~\mathrm{W}\) respectively. What will be the temperature on the new scale corresponding to a temperature of \(39^\circ ~\mathrm{C}\) on the Celsius scale?
1. \(78^\circ ~\mathrm{C}\)
2. \(117^\circ ~\mathrm{W}\)
3. \(200^\circ ~\mathrm{W}\)
4. \(139^\circ ~\mathrm{W}\)
The temperature of a body on the Kelvin scale is found to be x K. When it is measured by a Fahrenheit thermometer, it is found to be xF, then the value of x is:
1. 40
2. 313
3. 574.25
4. 301.25
Two rods, one made of aluminium and the other made of steel, having initial lengths \(l_1\) and \(l_2\) are connected together to form a single rod of length . The coefficient of linear expansion for aluminium and steel are and respectively. If the length of each rod increases by the same amount when their temperature is raised by \(t^\circ \mathrm{C},\) then the ratio \(\frac{l_1}{l_1+l_2}\) is:
1.
2.
3.
4.
Liquid oxygen at \(50\) K is heated up to \(300\) K at a constant pressure of \(1\) atm. The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 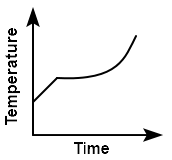 |
| 3. |  |
4. |  |
If the radius of a star is \(R\) and it acts as a black body, what would be the temperature of the star at which the rate of energy production is \(Q\)? \(\left(\sigma~ \text{is Stefan-Boltzmann constant}\right)\)
1. \(\frac{Q}{4\pi R^2\sigma}\)
2. \(\left(\frac{Q}{4\pi R^2\sigma}\right )^{\frac{-1}{2}}\)
3. \(\left(\frac{4\pi R^2 Q}{\sigma}\right )^{\frac{1}{4}}\)
4. \(\left(\frac{Q}{4\pi R^2 \sigma}\right)^{\frac{1}{4}}\)
Three rods made of the same material and having the same cross-section have been joined as shown in the figure. Each rod has the same length. The left and right ends are kept at \(0^{\circ}\text{C}~\text{and}~90^{\circ}\text{C},\) respectively. The temperature at the junction of the three rods will be:

1. \(45^{\circ}\text{C}\)
2. \(60^{\circ}\text{C}\)
3. \(30^{\circ}\text{C}\)
4. \(20^{\circ}\text{C}\)
Hot coffee in a mug cools from \(90^{\circ}\mathrm{C}\) to \(70^{\circ}\mathrm{C}\) in 4.8 minutes. The room temperature is \(20^{\circ}\mathrm{C}\). Applying Newton's law of cooling, the time needed to cool it further by \(10^{\circ}\mathrm{C}\) should be nearly:
| 1. | 4.2 minute | 2. | 3.8 minute |
| 3. | 3.2 minute | 4. | 2.4 minute |
One kilogram of ice at \(0^\circ \mathrm{C}\) is mixed with one kilogram of water at \(80^\circ \mathrm{C}.\) The final temperature of the mixture will be: (Take: Specific heat of water = \(4200\) J kg-1 K-1, latent heat of ice\(=336\) kJ kg-1)
| 1. | \(0^\circ \mathrm{C}\) | 2. | \(50^\circ \mathrm{C}\) |
| 3. | \(40^\circ \mathrm{C}\) | 4. | \(60^\circ \mathrm{C}\) |


