Mean kinetic energy per degree of freedom of gas molecules is:
1.
2. kT
3.
4.
PV versus T graphs of equal masses of \(H_2\), \(He\) and \(O_2\) are shown in the figure. Choose the correct alternative:
| 1. | A corresponds to \(H_2\), B to \(He\) and C to \(O_2\) | 2. | A corresponds to \(He\), B to \(H_2\), and C to \(O_2\) |
| 3. | A corresponds to \(He\), B to \(O_2\), and C to \(H_2\) | 4. | A corresponds to \(O_2\), B to \(He\) and C to \(H_2\) |
Molecular weight of two gases are \(M_1\) and \(M_2.\) At any temperature, the ratio of root mean square velocities \(v_1\) and \(v_2\) will be:
1. \(\sqrt{\frac{M_1}{M_2}}\)
2. \(\sqrt{\frac{M_2}{M_1}}\)
3. \(\sqrt{\frac{M_1+M_2}{M_1-M_2}}\)
4. \(\sqrt{\frac{M_1-M_2}{M_1+M_2}}\)
Which one of the following graph is correct at constant pressure?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Volume, pressure, and temperature of an ideal gas are \(V,\) \(P,\) and \(T\) respectively. If the mass of its molecule is \(m\), then its density is: [\(k\)=Boltzmann's constant]
| 1. | \(mkT\) | 2. | \(P \over kT\) |
| 3. | \(P \over kTV\) | 4. | \(Pm \over kT\) |
At what temperature is the root mean square speed of molecules of hydrogen twice as that at STP?
1. \(273\) K
2. \(546\) K
3. \(819\) K
4. \(1092\) K
At constant temperature, on increasing the pressure of a gas by 5% , its volume will decrease by:
1. 5%
2. 5.26 %
3. 4.26 %
4. 4.76 %
The curve between absolute temperature and \(\mathrm{v}^2_{rms}\) is:
| 1. |  |
2. |  |
| 3. |  |
4. | 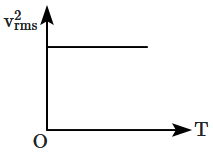 |
A thermally insulated piston divides a container into two compartments. Volume, temperature, and pressure in the right compartment are \(2V\), \(T\) and \(2P\), while in the left compartment the respective values are \(V\), \(T\) and \(P\). If the piston can slide freely, then in the final equilibrium position, the volume of the right-hand compartment will be:
| 1. | \(V\) | 2. | \(3V \over 5\) |
| 3. | \(9V \over 4\) | 4. | \(12V \over 5\) |
The pressure and temperature of two different gases are P and T with volume V for each. If they are mixed, keeping the same volume and temperature, the pressure of the mixture will be:
| 1. | P/2 | 2. | P |
| 3. | 2P | 4. | 4P |



