The mean free path for a gas, with molecular diameter \(d\) and number density \(n,\) can be expressed as:
1. \( \frac{1}{\sqrt{2} n \pi \mathrm{d}^2} \)
2. \( \frac{1}{\sqrt{2} n^2 \pi \mathrm{d}^2} \)
3. \(\frac{1}{\sqrt{2} n^2 \pi^2 d^2} \)
4. \( \frac{1}{\sqrt{2} n \pi \mathrm{d}}\)
1. \( \frac{1}{\sqrt{2} n \pi \mathrm{d}^2} \)
2. \( \frac{1}{\sqrt{2} n^2 \pi \mathrm{d}^2} \)
3. \(\frac{1}{\sqrt{2} n^2 \pi^2 d^2} \)
4. \( \frac{1}{\sqrt{2} n \pi \mathrm{d}}\)
The graph between volume and temperature in Charle's law is?
1. an ellipse
2. a circle
3. a straight line
4. a parabola
The ratio of the average translatory kinetic energy of He gas molecules to gas molecules is:
1.
2.
3.
4. 1
Without change in temperature, a gas is forced in a smaller volume. Its pressure increases because its molecules:
| 1. | strike the unit area of the container wall more often. |
| 2. | strike the unit area of the container wall at a higher speed. |
| 3. | strike the unit area of the container wall with greater force. |
| 4. | have more energy. |
If at a pressure of \(10^6\) dyne/cm2, one gram of nitrogen occupies \(2\times10^4\) c.c. volume, then the average energy of a nitrogen molecule in erg is:
| 1. | \(14\times10^{-13}\) | 2. | \(10\times10^{-12}\) |
| 3. | \(10^{6}\) | 4. | \(2\times10^{6}\) |
The translational kinetic energy of n moles of a diatomic gas at absolute temperature T is given by:
1.
2.
3.
4.
The translational kinetic energy of oxygen molecules at room temperature is 60 J. Their rotational kinetic energy will be?
1. 40 J
2. 60 J
3. 50 J
4. 20 J
The change in the internal energy of an ideal gas does not depend on?
| 1. | Number of moles |
| 2. | Change in temperature |
| 3. | Specific heat at constant pressure \(C_p\) of the gas |
| 4. | Specific heat at constant volume \(C_v\) of the gas |
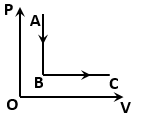
The figure shows a process for a gas in which pressure (P) and volume (V) of the gas change. If and are the molar heat capacities of the gas during the processes AB and BC respectively, then:
1.
2.
3.
4.
In the PV graph shown below for an ideal diatomic gas, the change in the internal energy is:
1.
2.
3.
4.