Which of the following diagrams (figure) depicts ideal gas behaviour?
| 1. | (a), (c) | 2. | (a), (d) |
| 3. | (c), (d) | 4. | (a), (b) |
Boyle's law is applicable for an:
| 1. | adiabatic process | 2. | isothermal process |
| 3. | isobaric process | 4. | isochoric process |
A cylinder containing an ideal gas is in a vertical position and has a piston of mass \(M\) that is able to move up or down without friction (figure). If the temperature is increased,

| 1. | both \(P\) and \(V\) of the gas will change. |
| 2. | only \(P\) will increase according to Charles' law. |
| 3. | \(V\) will change but not \(P.\) |
| 4. | \(P\) will change but not \(V.\) |
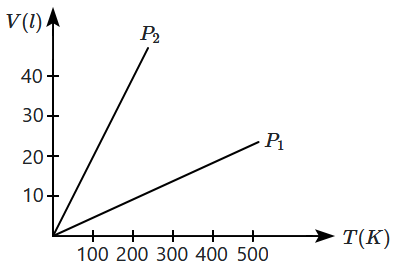
The volume versus temperature graphs for a given mass of an ideal gas are shown in the figure at two different values of constant pressure. What can be inferred about relation between \(\mathrm{P_1}\) and \(\mathrm{P_2}\)?
1. \(\mathrm{P_1}>\mathrm{P_2} \)
2. \(\mathrm{P_1}=\mathrm{P_2} \)
3. \(\mathrm{P_1}<\mathrm{P_2} \)
4. data is insufficient
(considering all gases to be ideal)
| 1. | same as the pressure initially. |
| 2. | \(2\) times the pressure initially. |
| 3. | \(10\) times the pressure initially. |
| 4. | \(20\) times the pressure initially. |
An inflated rubber balloon contains one mole of an ideal gas, has a pressure \(P,\) volume \(V\) and temperature \(T.\) If the temperature rises to \(1.1T,\) and the volume is increased to \(1.05V,\) the final pressure will be:
| 1. | \(1.1P\) |
| 2. | \(P\) |
| 3. | less than \(P\) |
| 4. | between \(P\) and \(1.1P\) |