If a gas undergoes the change in its thermodynamic state from A to B via two different paths, as shown in the given pressure (P) versus volume (V) graph, then:

1.
the temperature of the gas decreases in path 1 from A to B.
2.
the heat absorbed by the gas in path 1 is greater than in path 2.
3.
the heat absorbed by the gas in path 2 is greater than in path 1.
4.
the change in internal energy in path 1 is greater than in path 2.

The PV diagram of an ideal gas is shown in the figure. The work done by the gas in the process is given by:
| 1. | \(\frac{9}{2}P_0V_0\) | 2. | \(\frac{15}{2}P_0V_0\) |
| 3. | \(\frac{13}{2}P_0V_0\) | 4. | \(\frac{3}{2}P_0V_0\) |
A pressure \((P)\) versus volume \((V)\) graph is obtained for an ideal gas from state \(A\) to state \(B\) (as shown in the figure below). Which of the following is the correct statement?

| 1. | The temperature of gas increases continuously. |
| 2. | The temperature of gas decreases continuously. |
| 3. | The temperature first increases, then decreases. |
| 4. | The temperature first decreases, then increases. |
When the sink temperature is kept at 400 K, the efficiency of a Carnot engine is 50%. While keeping the source temperature constant, by how much should we reduce the sink temperature to increase the efficiency to 60%?
| 1. | 80 K | 2. | 70 K |
| 3. | 320 K | 4. | 240 K |
Which of the following graph shows the variation of pressure P with volume V for an ideal gas at a constant temperature?
| 1. |  |
2. |  |
| 3. |  |
4. | 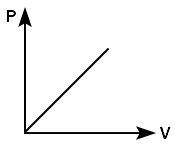 |
A gas performs the minimum work when it expands:
| 1. | Isochorically | 2. | Isobarically |
| 3. | Adiabatically | 4. | Isothermally |
An ideal gas goes from A to B via two processes, l and ll, as shown. If and are the changes in internal energies in processes I and II, respectively, then (\(P:\) pressure, \(V:\) volume)
| 1. | ∆U1 > ∆U2 | 2. | ∆U1 < ∆U2 |
| 3. | ∆U1 = ∆U2 | 4. | ∆U1 ≤ ∆U2 |
An ideal monoatomic gas \(\left(\gamma = \frac{5}{3}\right )\) absorbs 50 cal in an isochoric process. The increase in internal energy of the gas is:
| 1. | 20 cal | 2. | Zero |
| 3. | 50 cal | 4. | 30 cal |
The pressure-temperature (P-T) graph for two processes, A and B, in a system is shown in the figure. If and are work done by the gas in process A and B respectively, then:
| 1. | \(W_{1}\) = \(W_{2}\) | 2. | \(W_{1}\) < \(W_{2}\) |
| 3. | \(W_{1}\) > \(W_{2}\) | 4. | \(W_{1}\) = \(-W_{2}\) |
The variation of molar heat capacity at constant volume with temperature T for a monatomic gas is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |





