Isomerism exhibited by acetic acid and methyl formate is:
1. Functional
2. Chain
3. Geometrical
4. Central
Compounds with as molecular formula can exhibit-
1. Position isomerism
2. Metamerism
3. Functional isomerism
4. All of the above
Functional isomer is not possible for -
| 1. | Alcohols | 2. | Aldehydes |
| 3. | Alkyl halides | 4. | Cyanides |
Alkyl cyanide \(R-C \equiv N\) and alkyl isocyanides \(R- \overset{+}{N} \equiv C^-\) are:
| 1. | Tautomers | 2. | Metamers |
| 3. | Functional isomers | 4. | Geometrical isomers |
An isomer of ethanol is-
| 1. | Methanol | 2. | Diethyl ether |
| 3. | Acetone | 4. | Dimethyl ether |
The type of structural isomerism shown by given compounds is-
and 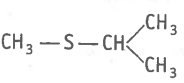
| 1. | Tautomerism | 2. | Positional isomerism |
| 3. | Functional isomerism | 4. | Ring Chain isomerism |
The pair of structures that does not represent isomers is:
| 1. | 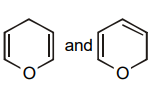 |
2. | 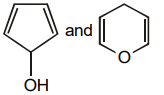 |
| 3. | 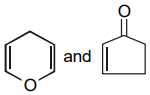 |
4. | 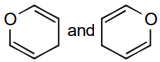 |
The number of isomeric structures for C2H7N would be:
1. 4
2. 3
3. 2
4. 1
Which of the following organic compounds exhibit tautomerism?
| I. | 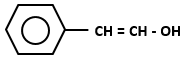 |
II. | 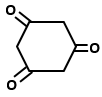 |
| III. | 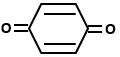 |
IV. | 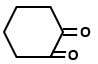 |
1. I and II
2. I, III and IV
3. I, II and IV
4. I, II, III and IV
The pair that represents chain isomers is-
| 1. | CH3CHCl2 and ClCH2CH2Cl | 2. | Propyl alcohol and Isopropyl alcohol |
| 3. | 2-Methylbutane and Neopentane | 4. | Diethyl ether and Dipropyl ether |


