If the electronic configuration of an element is , the four electrons that participate in the chemical bond formation will be from :
| 1. | 2. | ||
| 3. | 4. |
The electronic configurations of the elements A, B, and C are given below.
The stable form of A may be represented by the formula:
| 1. | A | 2. | A2 |
| 3. | A3 | 4. | A4 |
The electronic configurations of the elements A, B, and C are given below.
The bond between B and C will be :
| 1. | Ionic. | 2. | Covalent. |
| 3. | Hydrogen. | 4. | Coordinate. |
Most covalent compound among the following is :
| 1. | AlF3 | 2. | AlCl3 |
| 3. | AlBr3 | 4. | AlI3 |
The correct order of covalent bond character among
LiCl, BeCl2, BCl3 and CCl4, is:
1. LiCl < BeCl2 > BCl3 > CCl4
2. LiCl > BeCl2 < BCl3 < CCl4
3. LiCl < BeCl2 < BCl3 < CCl4
4. LiCl > BeCl2 > BCl3 > CCl4
Among the following, the maximum covalent character is shown by which compound:
1.
2.
3.
4.
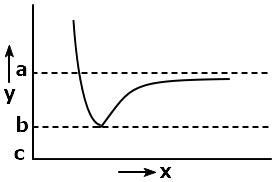
The following graph captures potential energy on the y-axis for hydrogen gas formation as a function of the internuclear distance on the x-axis:
The bond energy of H2 can be represented by-
| 1. | (c – a) | 2. | (b – a) |
| 3. | (c-a)/2 | 4. | (b-a)/2 |
Which of the following atomic orbitals would have the greatest overlap to form the strongest covalent bond?
| 1. | 2s-2s (σ) | 2. | 2s-2p (σ) |
| 3. | 2p-2p (π) | 4. | 2p-2p (σ) |