Activation energy of any chemical reaction can be calculated if one knows the value of
| 1. | Probability of collision. |
| 2. | Orientation of reactant molecules during collision. |
| 3. | Rate constant at two different temperatures. |
| 4. | Rate constant at standard temperature. |
Subtopic: Arrhenius Equation |
69%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Which plot of In k vs \(\frac{\text{I}}{\text{T}}\) is consistent with Arrhenius equation?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Arrhenius Equation |
66%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
The rate of a reaction quadruples when temperature changes from 27°C to 57°C. Calculate the energy of activation.
Given R = 8.314 J K–1 mol–1, log 4 = 0.6021
1. 380.4 kJ/mol
2. 3.80 kJ/mol
3. 3804 kJ/mol
4. 38.04 kJ/mol
Given R = 8.314 J K–1 mol–1, log 4 = 0.6021
1. 380.4 kJ/mol
2. 3.80 kJ/mol
3. 3804 kJ/mol
4. 38.04 kJ/mol
Subtopic: Arrhenius Equation |
52%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Effective collisions are known to possess:
A: Energy greater than threshold energy.
B: Breaking of old bond in reactant.
C: Formation of new bond in product.
D: High activation energy.
E: Proper orientation.
Choose the correct answer from the options given below:
1. A, B, C, D only
2. A, B, C, E only
3. A, C, D, E only
4. B, C, D, E only
A: Energy greater than threshold energy.
B: Breaking of old bond in reactant.
C: Formation of new bond in product.
D: High activation energy.
E: Proper orientation.
Choose the correct answer from the options given below:
1. A, B, C, D only
2. A, B, C, E only
3. A, C, D, E only
4. B, C, D, E only
Subtopic: Arrhenius Equation |
70%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Which of the following plot represents the variation of \(ln ~k\) versus \(\frac 1{T}\) in according with Arrhenius equation?
| 1. |  |
2. |
 |
| 3. |
 |
4. |
 |
Subtopic: Arrhenius Equation |
86%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Rate constants of a reaction at 500 K and 700 K are 0.04 \(s^{-1}\) and 0.14 \(s^{-1}\), respectively; then, activation energy of the reaction is:
(Given : log 3.5 = 0.5441, R = 8.31 J \(K^{-1} \) \(\mathrm {mol}^{-1}\))
1. 182310 J
2. 18500 J
3. 18219 J
4. 18030 J
(Given : log 3.5 = 0.5441, R = 8.31 J \(K^{-1} \) \(\mathrm {mol}^{-1}\))
1. 182310 J
2. 18500 J
3. 18219 J
4. 18030 J
Subtopic: Arrhenius Equation |
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
In the light of the above statements choose the correct answer from the options given below:
| Assertion (A): | A reaction can have zero activation energy. |
| Reason (R): | The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to threshold value, is called activation energy. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Arrhenius Equation |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The plot of ln k vs \({1 \over T}\) for the following reaction
\(2N_2O_5(g) \rightarrow 4NO_2 (g) + O_2(g) \) gives a straight line with the slope of the line equal to \(-1.0 \times 10^4 K \).
The activation energy for the reaction in J mol–1 is:
(Given R = 8.3 J K–1 mol–1)
| 1. | \(4.0 \times 10^2 \) | 2. | \(4.0 \times 10^{-2} \) |
| 3. | \(8.3 \times 10^{-4} \) | 4. | \(8.3 \times 10^4 \) |
Subtopic: Arrhenius Equation |
76%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
For a reaction AB, enthalpy of reaction is and enthalpy of activation is . The correct potential energy profile for the reaction is:
| 1. | 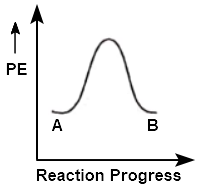 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Arrhenius Equation |
71%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The slope of Arrhenius Plot (ln k v/s ) of the first-order reaction is . The value of Ea of the reaction is:
[Given R = 8.314 JK–1 mol–1]
| 1. | 166 kJ mol–1 | 2. | –83 kJ mol–1 |
| 3. | 41.5 kJ mol–1 | 4. | 83.0 kJ mol–1 |
Subtopic: Arrhenius Equation |
73%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


