The number of primary carbon atoms in the following compound are:

1.
6
2.
2
3.
4
4.
3

Isomerism exhibited by acetic acid and methyl formate is:
1. Functional
2. Chain
3. Geometrical
4. Central
0.2 g of an organic compound on complete combustion produces 0.44 g of CO2. The percentage of carbon is -
1. 50%
2. 60%
3. 70%
4. 80%
The Prussian blue colour obtained during the test of nitrogen by Lassaigne's test is due to the formation of-
| 1. | Fe4[Fe(CN)6]3 | 2. | Na3[Fe(CN)6] |
| 3. | Fe(CN)3 | 4. | Na4[Fe(CN)5NOS] |
The most stable carbocation among the following is:
| 1. |  |
2. |  |
| 3. | 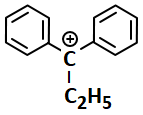 |
4. |  |
The arrangement in decreasing order of stability of H3, H5,(CH3)2H and (CH3)3 free radicals is-
1. H3 > H5 > (CH3)2H > (CH3)3
2. (CH3)3 > (CH3)2H > H5 > H3
3. H5 > H3 > (CH3)2H > (CH3)3
4. (CH3)3 > (CH3)2H > H3 >H5
The optically active compound among the following is-
| 1. | Glycerine | 2. | Acetaldehyde |
| 3. | Glyceraldehyde | 4. | Acetone |
In Duma's method of estimation of nitrogen, 0.35 g of an organic compound gave 55 ml of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound would be:
(Aqueous tension at 300 K = 15 mm)
1. 16.45
2. 27.45
3. 44.45
4. 35.45
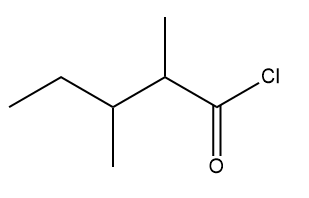
The IUPAC name of the above mentioned compound is -
1. 3, 4-Dimethylpentanoyl chloride
2. 1-Chloro-1-oxo-2,3-dimethylpentane
3. 2-Ethyl-3-methylbutanoylchloride
4. 2, 3-Dimethylpentanoyl chloride
The best method used for the separation of naphthalene and benzoic acid from their mixture is -
1. Sublimation
2. Chromatography
3. Crystallisation
4. Distillation


