Isomerism exhibited by acetic acid and methyl formate is:
1. Functional
2. Chain
3. Geometrical
4. Central
Which types of isomerism can be exhibited by compounds with the molecular formula C4H11N?
1. Position isomerism
2. Metamerism
3. Functional isomerism
4. All of the above
Functional isomer is not possible for:
1. Alcohols
2. Aldehydes
3. Alkyl halides
4. Cyanides
Alkyl cyanide \(R-C \equiv N\) and alkyl isocyanides \(R- \overset{+}{N} \equiv C^-\) are:
| 1. | Tautomers | 2. | Metamers |
| 3. | Functional isomers | 4. | Geometrical isomers |
Which of the following is an isomer of ethanol?
| 1. | Methanol | 2. | Diethyl ether |
| 3. | Acetone | 4. | Dimethyl ether |
The type of structural isomerism shown by given compounds is-
and 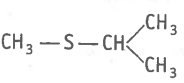
| 1. | Tautomerism | 2. | Positional isomerism |
| 3. | Functional isomerism | 4. | Ring Chain isomerism |
The pair of structures that does not represent isomers is:
| 1. | 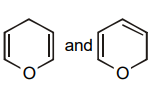 |
2. | 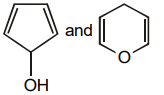 |
| 3. | 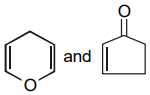 |
4. | 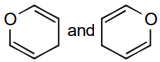 |
The number of isomeric structures for C2H7N would be:
1. 4
2. 3
3. 2
4. 1
Which of the following organic compounds exhibit tautomerism?
| I. | 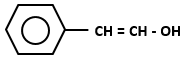 |
II. | 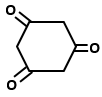 |
| III. | 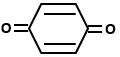 |
IV. | 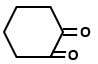 |
1. I and II
2. I, III and IV
3. I, II and IV
4. I, II, III and IV
The pair that represents chain isomers is-
| 1. | CH3CHCl2 and ClCH2CH2Cl | 2. | Propyl alcohol and Isopropyl alcohol |
| 3. | 2-Methylbutane and Neopentane | 4. | Diethyl ether and Dipropyl ether |






