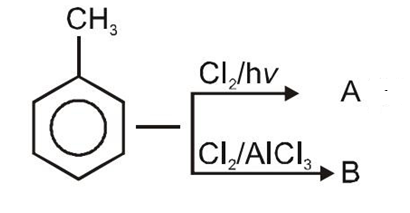
The major product obtained in the given reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The mechanism & intermediate involved in the above reaction are:
1. Aromatic electrophilic substitution & carbocation
2. Aromatic Nucleophilic substitution & carbanion
3. Aromatic free radical substitution & Free radical
4. Carbene based substitution reaction & Carbene
A compound among the following that does not undergo Friedel-Craft's reaction easily is-
1. Cumene
2. Xylene
3. Nitrobenzene
4. Toluene
The compounds among the following that can be generated by Friedel craft acylation is/are -
| I. | 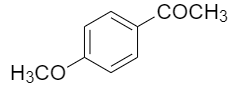 |
| II. |  |
| III. |  |
| IV. |  |
| 1. | II, III and IV | 2. | I, III and IV |
| 3. | I and II | 4. | II and III |
Cyclopentadiene is much more acidic than cyclopentane, beacuse -
1. Cyclopentadiene has conjugated double bonds.
2. Cyclopentadiene has both sp2 and sp3 hybridized carbon atoms.
3. Cyclopentadiene is a strain-free cyclic system.
4. Cyclopentadienyl anion ion, the conjugate base of cyclopentadiene, is an aromatic species and hence has higher stability.
A compound among the following that gives the maximum % of meta product on nitration
(using HNO3/H2SO4) is :
1. Toluene
2. Aniline
3. Benzene
4. Isopropyl benzene
The major product in the following reaction is-
| 1. | 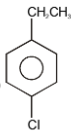 |
2. | 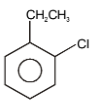 |
| 3. | 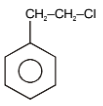 |
4. | 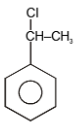 |
The D in the above-mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The product B in the above-mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Consider the following reaction:-
A and B are isomeric compounds. Additionally, A is a monochloro derivative. These two can be differentiated easily by using:
1. AgN(aq)
2.
3. Conc.HN/
4. NaCl(aq)