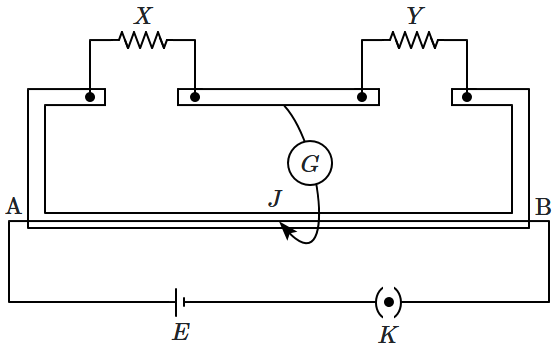
In a meter bridge experiment, the null point is at a distance of \(30~\text{cm}\) from \(\mathrm{A}\). If a resistance of \(16~\Omega\) is connected in parallel with resistance \(Y\), the null point occurs at \(50~\text{cm}\) from \(\mathrm{A}\). The value of the resistance \(Y\) is:
1.
\(\dfrac{112}{3}~\Omega\)
2.
\(\dfrac{40}{3}~\Omega\)
3.
\(\dfrac{64}{3}~\Omega\)
4.
\(\dfrac{48}{3}~\Omega\)

The temperature at which the rms speed of atoms in neon gas is equal to the rms speed of hydrogen molecules at \(15^{\circ} \mathrm{C}\) is: (Atomic mass of neon \(=20.2\) u, molecular mass of hydrogen \(=2\) u)
| 1. | \(2.9\times10^{3}\) K | 2. | \(2.9\) K |
| 3. | \(0.15\times10^{3}\) K | 4. | \(0.29\times10^{3}\) K |
Two planets are in a circular orbit of radius \(R\) and \(4R\) about a star. At a specific time, the two planets and the star are in a straight line. If the period of the closest planet is \(T,\) then the star and planets will again be in a straight line after a minimum time:
2. \((4)^{\frac13}T\)
3. \(2T\)
4. \(8T\)
The correct order for boiling points of the following compounds is:
| 1. | AsH3 > PH3 > NH3 > SbH3 > BiH3 |
| 2. | BiH3 > SbH3 > NH3 > AsH3 > PH3 |
| 3. | NH3 > PH3 > AsH3 > SbH3 > BiH3 |
| 4. | PH3 > NH3 > AsH3 > SbH3 > BiH3 |
Match List-I with List-II:
| List-I | List-II | ||
| (a) | 4.48 litres of O2 at STP | (i) | 0.2 moles |
| (b) | 12.022 × 1022 molecules of H2O | (ii) | 12.044 × 1023 molecules |
| (c) | 96 g of O2 | (iii) | 6.4 g |
| (d) | 88 g of CO2 | (iv) | 67.2 litres at STP |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (i) | (iii) | (iv) | (ii) |
| 2. | (iii) | (i) | (iv) | (ii) |
| 3. | (iv) | (i) | (ii) | (iii) |
| 4. | (iii) | (i) | (ii) | (iv) |
Given below are two statements:
| Statement I: | Aldehydes and ketones having at least one \(\alpha\)-hydrogen undergo aldol condensation in the presence of dilute alkali as catalyst. |
| Statement II: | When aldol condensation is carried out between two different aldehydes, it is called cross aldol condensation. Ketones do not give this reaction. |
In light of the above statements, choose the most appropriate answer from the options given below:
| 1. | Statement I is correct and Statement II is incorrect. |
| 2. | Statement I is incorrect and Statement II is correct. |
| 3. | Both Statement I and Statement II are correct. |
| 4. | Both Statement I and Statement II are incorrect. |
Match List-I with List-II:
| List-I Elements |
List-II Atomic radii (pm) |
||
| (a) | O | (i) | 88 |
| (b) | C | (ii) | 74 |
| (c) | B | (iii) | 66 |
| (d) | N | (iv) | 77 |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (iv) | (iii) | (ii) | (i) |
| 3. | (i) | (iv) | (iii) | (ii) |
| 4. | (iii) | (iv) | (i) | (ii) |
The correct reaction among the following is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The number of bridging carbonyl groups in [Co2(CO)8] and [Mn2(CO)10], respectively are:
| 1. | 2 and 0 | 2. | 2 and 2 |
| 3. | 2 and 4 | 4. | 0 and 2 |
LiF is sparingly soluble in water because it has:
1. partial covalent character.
2. small electronegativity.
3. high lattice enthalpy.
4. low hydration enthalpy.



