The \(x\text-t\) graph of a particle performing simple harmonic motion is shown in the figure. The acceleration of the particle at \(t=2\) s is:

1. \(-\dfrac{\pi^2}{16} ~\text{ms}^{-2}\)
2. \(\dfrac{\pi^2}{8}~ \text{ms}^{-2}\)
3. \(-\dfrac{\pi^2}{8} ~\text{ms}^{-2}\)
4. \(\dfrac{\pi^2}{16} ~\text{ms}^{-2}\)
Subtopic: Simple Harmonic Motion |
60%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The mass of CO2 produced by heating 20 g of 20% pure limestone as per the given below equation is:
\(\left[\mathrm{CaCO}_3 \stackrel{1200 \mathrm{~K}}{\longrightarrow} \mathrm{CaO}+\mathrm{CO}_2\right] \)
\(\left[\mathrm{CaCO}_3 \stackrel{1200 \mathrm{~K}}{\longrightarrow} \mathrm{CaO}+\mathrm{CO}_2\right] \)
| 1. | 1.32 g | 2. | 1.12 g |
| 3. | 1.76 g | 4. | 2.64 g |
Subtopic: Equation Based Problem |
72%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Some tranquilizers are listed below. Which one from the following belongs to barbiturates?
| 1. | Veronal | 2. | Chlordiazepoxide |
| 3. | Meprobamate | 4. | Valium |
Subtopic: Classification of Medicines |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given below are two statements: one is labeled as Assertion (A) and the other is labeled as Reason (R):
In light of the above statements, choose the correct answer from the options given below:
| Assertion (A): | Helium is used to dilute oxygen in the diving apparatus. |
| Reason (R): | Helium has a high solubility in O2. |
| 1. | (A) is False but (R) is True. |
| 2. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 3. | Both (A) and (R) are True and (R) is not the correct explanation of (A). |
| 4. | (A) is True but (R) is False. |
Subtopic: Concentration Terms & Henry's Law |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
In the light of the above statements choose the correct answer from the options given below:
| Assertion (A): | Metallic sodium dissolves in liquid ammonia giving a deep blue solution, which is paramagnetic. |
| Reason (R): | The deep blue solution is due to the formation of amide. |
| 1. | (A) is false but (R) is true. |
| 2. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 3. | Both (A) and (R) are true and R is NOT the correct explanation of (A). |
| 4. | (A) is true but (R) is false. |
Subtopic: Reasoning Questions |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The correct order of energies of molecular orbitals of N2 molecule is:
| 1. | \( \sigma \text { 1s }<\sigma^* \text { 1s }<\sigma 2 s<\) \(\sigma^* 2 s<\left(\pi 2 p_x=\pi 2 p_y\right)<\) \(\left(\pi^* 2 p_x=\pi^* 2 p_y\right)<\sigma 2 p_z<\sigma^* 2 p_z\) |
| 2. | \( \sigma \text { 1s }<\sigma^* \text { 1s }<\sigma 2 s<\sigma^* 2 s<\) \(\left(\pi 2 p_x=\pi 2 p_y\right)< \) \(\sigma 2 p_z<\left(\pi^* 2 p_x=\pi^* 2 p_y\right)<\sigma^* 2 p_z \) |
| 3. | \( \sigma \text { 1s }<\sigma^* \text { 1s }<\sigma 2 s<\sigma^* 2 s<\sigma 2 p_z<\) \( \left(\pi 2 p_x=\pi 2 p_y\right)<\) \(\left(\pi^* 2 p_x=\pi^* 2 p_y\right)<\sigma^* 2 p_z \) |
| 4. | \( \sigma \text { 1s }<\sigma^* \text { 1s }\) \(<\sigma 2 s<\sigma^* 2 s<\sigma 2 p_z< \) \( \sigma^* 2 p_z<\left(\pi 2 p_x=\pi 2 p_y\right)<\) \(\left(\pi^* 2 p_x=\pi^* 2 p_y\right) \) |
Subtopic: M.O.T |
70%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
In the light of the above statements choose the correct answer from the options given below:
| Assertion (A): | A reaction can have zero activation energy. |
| Reason (R): | The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to threshold value, is called activation energy. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Arrhenius Equation |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Weight (g) of two moles of the organic compound, which is obtained by heating sodium ethanoate with sodium hydroxide in the presence of calcium oxide is:
1. 18
2. 16
3. 32
4. 30
1. 18
2. 16
3. 32
4. 30
Subtopic: Carboxylic Acids: Preparation & Properties |
61%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given is a reaction for your reference:
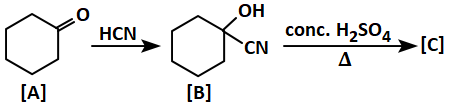
The final product [C] is:

The final product [C] is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties |
61%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Select the correct statements from the following:
Choose the correct answer from the option given below:
| A: | Atoms of all elements are composed of two fundamental particles. |
| B: | The mass of the electron is \(9.10939 \times 10^{-31} \mathrm{~kg} \text {. }\). |
| C: | All the isotopes of a given element show same chemical properties. |
| D: | Protons and electrons are collectively known as nucleons. |
| E: | Dalton's atomic theory regarded the atom as an ultimate particle of matter. |
| 1. | B, C and E only | 2. | A, B and C only |
| 3. | C, D and E only | 4. | A and E only |
Subtopic: Introduction |
71%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.


