Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
Assertion (A):
A reaction can have zero activation energy.
Reason (R):
The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to threshold value, is called activation energy.
In the light of the above statements choose the correct answer from the options given below:
1.
Both (A) and (R) are true and (R) is the correct explanation of (A).
2.
Both (A) and (R) are true but (R) is not the correct explanation of (A).
3.
(A) is true but (R) is false.
4.
(A) is false but (R) is true.
Subtopic: Arrhenius Equation |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Weight (g) of two moles of the organic compound, which is obtained by heating sodium ethanoate with sodium hydroxide in the presence of calcium oxide is:
1. 18
2. 16
3. 32
4. 30
1. 18
2. 16
3. 32
4. 30
Subtopic: Carboxylic Acids: Preparation & Properties |
61%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given is a reaction for your reference:
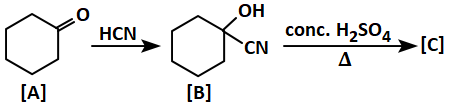
The final product [C] is:

The final product [C] is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties |
61%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Select the correct statements from the following:
Choose the correct answer from the option given below:
| A: | Atoms of all elements are composed of two fundamental particles. |
| B: | The mass of the electron is \(9.10939 \times 10^{-31} \mathrm{~kg} \text {. }\). |
| C: | All the isotopes of a given element show same chemical properties. |
| D: | Protons and electrons are collectively known as nucleons. |
| E: | Dalton's atomic theory regarded the atom as an ultimate particle of matter. |
| 1. | B, C and E only | 2. | A, B and C only |
| 3. | C, D and E only | 4. | A and E only |
Subtopic: Introduction |
71%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The stability of Cu2+ is greater than that of Cu+ salts in an aqueous solution due to:
1. Second ionisation enthalpy
2. First ionisation enthalpy
3. Enthalpy of atomization
4. Hydration energy
1. Second ionisation enthalpy
2. First ionisation enthalpy
3. Enthalpy of atomization
4. Hydration energy
Subtopic: d-Block Elements- Properties & Uses |
70%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The relation between \(n_m \) (\(n_m \) = number of permissible values of magnetic quantum number (ml) for a given value of azimuthal quantum number (l) is:
1. \(n_m = l+2\)
2. \(l = {n_m -1 \over 2}\)
3. \(l= 2n_m +1\)
4. \(n_m = 2l^2 + 1 \)
1. \(n_m = l+2\)
2. \(l = {n_m -1 \over 2}\)
3. \(l= 2n_m +1\)
4. \(n_m = 2l^2 + 1 \)
Subtopic: Shell & Subshell |
67%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Intermolecular forces are forces of attraction and repulsion between interacting particles that will be:
Choose the most appropriate answer from the options given below:
1. A, C, D, E are correct
2. B, C, D, E are correct
3. A, B, C, D, E are correct
4. A, B, C, E are correct
| A. | dipole - dipole forces |
| B. | dipole-induced dipole forces |
| C. | hydrogen bonding |
| D. | covalent bonding |
| E. | dispersion forces |
1. A, C, D, E are correct
2. B, C, D, E are correct
3. A, B, C, D, E are correct
4. A, B, C, E are correct
Subtopic: Introduction to States of Matter |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Homoleptic complex from the following complexes is:
1. Triamminetriaquachromium (III)
2. Potassium trioxalatoaluminate (III)
3. Diamminechloridonitrito-N-platinum (II)
4. Pentaamminecarbonatocobalt (III) chloride
1. Triamminetriaquachromium (III)
2. Potassium trioxalatoaluminate (III)
3. Diamminechloridonitrito-N-platinum (II)
4. Pentaamminecarbonatocobalt (III) chloride
Subtopic: Introduction, Classification and Nomenclature |
77%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Among the given reactions, which one does not result in the formation of a primary amine as the product?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Amines - Preparation & Properties |
71%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Which one of the following statements is correct?
| 1. | Mg plays role in neuromuscular function and interneuronal transmission. |
| 2. | The daily requirement of Mg and Ca in the human body is estimated to be 0.2-0.3 g. |
| 3. | All enzymes that utilize ATP in phosphate transfer require Ca as the cofactor. |
| 4. | The bone in the human body is an inert and unchanging substance. |
Subtopic: Biological Importance of S Block Elements and & Its Ore |
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.


