Isomers of a substance must have the same-
1. Structure
2. Physical properties
3. Chemical properties
4. Molecular formula
But-2-ene exhibits cis-trans-isomerism due to
1. rotation around C2-C3 double bond
2. rotation around C3-C4 sigma bond
3. rotation around C1-C2 bond
4. restricted rotation around C=C bond
Which of the following compounds is not chiral ?
1. DCH2CH2CH2Cl
2. CH3CH2CHDCl
3. CH3CHDCH2Cl
4. CH3CHClCH2D
Which of the following compounds will exhibit cis-trans (geometrical) isomerism?
1. but-2-ene
2. Butanol
3. but-2-yne
4. buten-2-ol
An alkane can form Structural isomers if the number of carbon atoms is-
1. From 1 carbon atom onwards
2.
3. From 2 carbon atoms onwards
4.
Which of the following molecules exhibits chirality?
1. 2-Methylhexane
2. 3-Methylhexane
3. Neopentane
4. Isopentane
The property by virtue of which a compound can turn the plane of polarization of light is known as:
1. Photolysis
2. Phosphorescence
3. Optical activity
4. Polarization
The compound which exhibits optical isomerism is:
1. CH3CHOHCH3
2. (CH3)2CHCH2CH3
3. CH3CHClCH2CH3
4. CH3CCl2CH2CH3
The total number of isomeric carbocations possible for the formula C4H9+ is:
(1) 3
(2) 4
(3) 2
(4) 5
Only two isomeric monochloro derivatives are possible for:
(1) n-butane
(2) 2,4-dimethylpentane
(3) benzene
(4) 2-methylbutane
Which of the following acids shows stereoisomerism?
1. Oxalic acid
2. Tartaric acid
3. Acetic acid
4. Formic acid
A racemic mixture is a mixture of:
1. meso and its isomers
2. d- and its l-isomers in equal proportions
3. d- and l-isomers in different proportions
4. meso and d-isomers
Compounds which rotate plane polarised light clockwise direction are known as:
1. Dextrorotatory
2. Laevorotatory
3. Optically inactive compounds
4. Racemic mixture
Two possible stereo-structures of CH3CHOH.COOH, that are optically active, called:
| 1. | Diastereomers | 2. | Atropisomers |
| 3. | Enantiomers | 4. | Mesomers |
The change in optical rotation with time of freshly prepared solution of sugar is known as–
1. Specific rotation
2. Inversion
3. Rotation
4. Mutarotation
Which of the following amino acids does not have an L-enantiomer?
1. Cysteine
2. Threonine
3. Glutamic acid
4. Glycine
The isomerism exhibited by the given molecules
| CH3-CH2-CH2-OH | 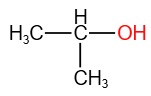 |
| (I) | (II) |
| 1. | Chain isomerism | 2. | Position isomerism |
| 3. | Both (1) & (2) | 4. | Metamerism |
The total number of isomers possible for the molecular formula C₇H₈O are:
1. 3
2. 4
3. 5
4. 6
Which of the following compounds exhibit stereoisomerism?
1. 2-methylbut-1-ene
2. 3-methylbut-1-ene
3. 3-methylbutanoic acid
4. 2-methylbutanoic acid
Which of the following have only one type of carbon atoms?
| 1. | 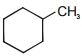 |
2. | 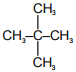 |
| 3. | 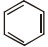 |
4. | 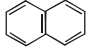 |
Dihedral angle in staggered form of ethane is:
1.
2.
3.
4.
Which of the following compounds can show tautomerism?
| 1. | 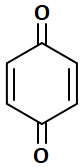 |
2. | 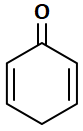 |
| 3. | 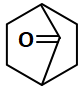 |
4. | 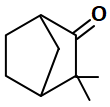 |
1. I, II, III, IV
2. I and II
3. III and IV
4. Only II
 |
 |
| I | II |
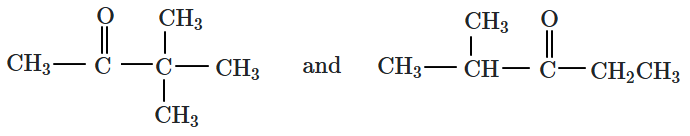
| 1. | Position isomers | 2. | Chain isomers |
| 3. | Stereoisomers | 4. | Tautomers |
| Column I (Structure of compounds) |
Column II (Types of isomerism) |
||
| A. | CH3CH2CH2CH2CH3 and |
I. | Chain isomerism |
| B. | CH3CH2CH2OH and |
II. | Position isomerism |
| C. | 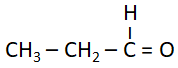 |
III. | Metamerism |
| D. | CH3OC3H7 and C2H5OC2H5 | IV. | Functional group isomerism |
| A | B | C | D | |
| 1. | I | II | III | IV |
| 2. | II | III | I | IV |
| 3. | IV | I | II | III |
| 4. | I | II | IV | III |
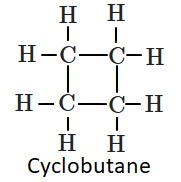
| I: | 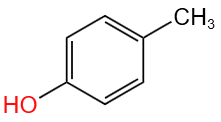 |
| II: | 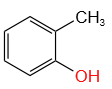 |
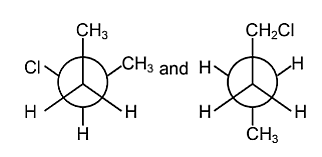
What is the relation between the Newmans' projections given below?
| 1. | 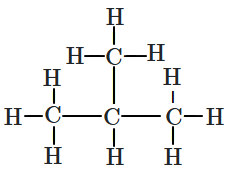 |
2. | 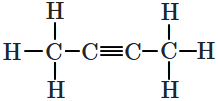 |
| 3. | 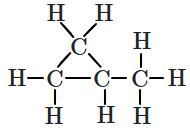 |
4. | 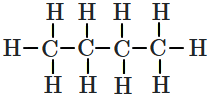 |
| 1. | \(C-C\) | 2. | \(C-O\) |
| 3. | \(C-H\) | 4. | \(O-H\) |