The linear molecule among the following is
1.
2.
3.
4.
The molecule having the highest dipole moment is
1.
2.
3.
4.
The species which has triangular planar geometry is
1.
2.
3.
4.
The correct sequence of stability for the given molecular species (from the given options) is:
1.
2.
3.
4.
From the given structures, the correct structure(s) of is are
l. 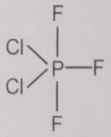
ll. 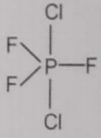
lll. 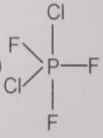
1. Only l
2. Only ll
3. Only lll
4. l, ll and lll
The pair that is isostructural (i.e. having the same shape and hybridization) is
1.
2.
3.
4.
Which of the following represents the correct order of dipole moment?
1.
2.
3.
4.
Which of the following has the shortest bond length?
1.
2.
3.
4.
Assuming that Hund's rule is violated by the diatomic molecule , its bond order and magnetic nature will be respectively
1. 1, diamagnetic
2. 1, paramagnetic
3. 2, diamagnetic
4. 2, paramagnetic






