A: Resonating structures should have the same number of unpaired electrons.
R: The energy content of all resonating structures is the same.
1. Both Assertion & Reason are true and the reason is the correct explanation of the assertion.
2. Both Assertion & Reason are true but the reason is not the correct explanation of the assertion.
3. Assertion is a true statement but Reason is false.
4. Both Assertion and Reason are false statements.
| Assertion (A): | Cyclopentadienyl anion is much more stable than the allyl anion. |
| Reason (R): | Cyclopentadienyl anion is aromatic while allyl anion is only resonance stabilized. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A) |
| 2. | Both (A) and (R) are True but (R) does not correctly explain (A) |
| 3. | (A) is True but (R) is False |
| 4. | Both (A) and (R) are False |
| Assertion (A): | Benzene and naphthalene are aromatic hydrocarbons. |
| Reason (R): | Benzene and naphthalene contain the same number of delocalized π electrons. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is a True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Given below are two statements:
| Statement I: | Alkyl groups show +l effect. |
| Statement II: | Halogens are -l and +R groups. |
| 1. | Statement I is incorrect and Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct, and Statement II is incorrect. |
| Assertion (A): |
2,3-Dimethyl-2-butene is the most stable alkene due to 12- hydrogen atoms. |
| Reason (R): | 'H' is more electronegative than carbon. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False |
| 4. | Both (A) and (R) are False. |
Given below two statements:
| Assertion(A): | A compound having a non-superimposable mirror image is optically active. |
| Reason(R): | Optically active compound must contain chiral carbon. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | Both (A) and (R) are True but (R) does not correctly explain (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
The most stable carbanion intermediate in the following is:
| 1. |  |
2. |  |
| 3. |  |
4. | All of these are equally stable. |
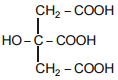
The IUPAC name of the above mentioned compound is -
1. Citric acid
2. 3-Hydroxy pentane-1,5-dioic acid
3. 2-Hydroxypropane-1,2,3-tricarboxylic acid
4. 2-Carboxy-2-hydroxy propane-1,3-dicarboxylic acid
The pair that represents chain isomers is :-
| 1. |  |
 |
| 2. |  |
 |
| 3. |  |
 |
| 4. | 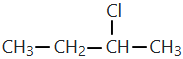 |
 |
The pair among the following that does not contain position isomers is -
| 1. | 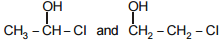 |
| 2. | 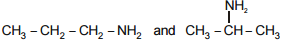 |
| 3. |  |
| 4. |  |






