The molecule that exhibits non-planarity is:
1.

2.

3.

4.





The aromatic compound among the following is:-
| 1. | 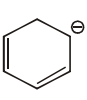 |
2. |  |
| 3. | 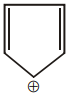 |
4. | 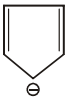 |
Which carbocation is the most stable among the options provided?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The presence of a halogen on the benzene ring in a nitration reaction leads to:
| 1. | Direct nitro group to come at meta and deactivate the ring due to –I effect of halogen. |
| 2. | Direct nitro group to come at ortho and para position and deactivate the ring due to –I effect of halogen. |
| 3. | Direct nitro group to come at meta and activate the ring toward nitration reaction. |
| 4. | Nitration reaction does not take place due to deactivation caused by –I effect of halogen. |
The pair of structures that does not represent isomers is:
| 1. | 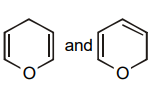 |
2. | 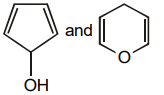 |
| 3. | 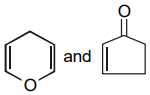 |
4. | 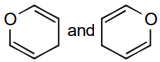 |
Which among these can exhibit tautomerism?
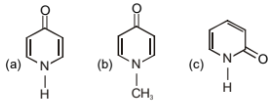
1. a only
2. b only
3. c only
4. a and c
Total number of stereoisomers of the compound:-
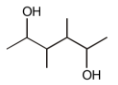
1. 10
2. 8
3. 6
4. 4
The effect that makes 2,3–dimethyl-2-butene more stable than 2-butene is-
1. Resonance
2. Hyperconjugation
3. Steric effect
4. Inductive effect

The IUPAC name of the above mentioned compound is -
1. Cyclohexylidenemethanone
2. Cyclohexylidemethanone
3. Cyclohexylidenylmethanone
4. Cyclohexdenemethanone
Which of the following compound requires minimum energy for free rotation across double bond between ring :
| 1. |  |
2. |  |
| 3. |  |
4. |  |






