The IUPAC name of the compound is :
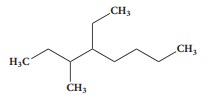
1. 2,3 - Diemethylheptane
2. 3- Methyl -4- ethyloctane
3. 5-Ethyl -6-methyloctane
4. 4-Ethyl -3 - methyloctane.

In the reaction,
a chiral centre is produced. This product would be
1. racemic mixture
2. meso compound
3. dextrorotatory
4. laevorotatory
The compound that contains only sp³ hybridized carbon atoms is:
1. HCOOH
2.
3.
4.
How many distinct products can be formed from the monochlorination of 2-methylbutane, excluding optical isomers?
| 1. | 2 | 2. | 3 |
| 3. | 4 | 4. | 5 |
Which of the following is the fundamental requirement for a molecule to exhibit optical activity?
1. Presence of at least one asymmetric carbon atom
2. Presence of a plane of symmetry
3. Non-superimposability of the molecule on its mirror image
4. The ability to rotate polarized light to the right only
The \(C - H\) bond distance is longer in -
| 1. | \(C_2H_2\) | 2. | \(C_2H_4\) |
| 3. | \(C_2H_6\) | 4. | \(C_2H_2Br_2\) |
The number of structural isomers of that are ethers is/are :
1. 1
2. 2
3. 3
4. 4
Which of the following has a zero dipole moment?
1. 1-butene
2. cis-2-butene
3. trans-2-butene
4. 2 methyl-1-propene
The total number of isomers of are-
| 1. | 8 | 2. | 7 |
| 3. | 6 | 4. | 5 |
How many stereoisomers are there for tartaric acid?
1. 3
2. 4
3. 5
4. 0






