In a body-centred cubic arrangement, A ions occupy the centre while B ions occupy the corners of the cube. The formula of the solid is
1. AB
2. AB2
3. A2B
4. AB3
If three elements A, B and C crystallized in cubic solid lattice with A atoms at corners, B atoms at cube centres and C atoms at the edges, the formula of the compound is
1. ABC
2. ABC3
3. AB3C
4. A3B2C3
In a cubic unit cell, seven of the eight corners are occupied by atom A and having of faces are occupied of B. The general formula of the substance having this type structure would be
1. A7B14
2. A14B7
3. A7B24
4. A9B24
In a cubic closed packed structure of mixed oxides, the lattice is made up of oxide ions, 20% of tetrahedral Voids are occupied by divalent A2+ ions and 50% of the octahedral voids by trivalent B3+. The formula of the oxide is?
1. A4B5O10
2. A2B5O5
3. A2BO
4. A4B5O8
A compound formed by elements A and B crystallizes in cubic structure where A atoms are at the corners or a cube and B atoms are at the face centre. The formula of the compound is :
1. AB
2. AB2
3. AB3
4. AB4
The radius of Ag+ ion is 126 pm while that of I- ion is 216 pm. The coordination number of Ag in AgI is
1. 8
2. 6
3. 4
4. 2
Which of the following crystals have 6:6 coordination?
1. MnO
2. NH4I
3. ZnS
4. none of these
TiO2 (rutile) shows 6:3 coordination. The solid having rutile like structure among the following is
1. KCl
2. SnO2
3. ZnS
4. none of these
A solid having unit cell made up of planes as shown in figure coordination number of 'X' is
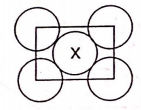
1. 12
2. 10
3. 6
4. 4
A compound contains two types of atoms: X and Y. It crystallizes in a cubic lattice with atoms X at the corners of the unit cell and atoms Y at the body centres.
The simples possible formula of this compound is:
1. XY
2. X2Y2
3. XY6
4. X8Y






