The closest distance between two atoms (in terms of edge length) would be highest for which of unit cell, assuming the edge length of each unit cell be 'a'
1. FCC unit cell
2. BCC unit cell
3. diamond unit cell
4. primitive cubic cell
A X+ Y- has the Y- ions arranged as below. If the X+ ions occupy half of the tetrahedral sites in the structure. The formula of solid is
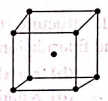
1. XY
2. X3Y4
3. XY2
4. X2Y
A solid XY has NaCl structure. If radius of X+ is 100 pm, the radius of Y- ion is
1. 136.6 to 241.6 pm
2. 341.6 to 134.6 pm
3. 128 pm
4. 136.8 pm
The ionic radii of Rb+ and I- are 1.46 Å and 2.16 Å respecitvely. The most probable type of structure exhibited by it is
1. CaF2 type
2. ZnS type
3. CsCl type
4. NaCl type
A solid has 3 types of atoms namely X, Y and Z. X forms an FCC lattice with Y atoms occupying all the tetrahedral voids and Z atoms occupying half the octahedral voids. The formula of the solid is
1. XYZ
2. X2Y4Z
3. X4YZ2
4. X4Y2Z
Calculate the of X-rays which give a diffraction angle 2 for a crystal. (Given interplanar distance=0.200 nm; diffraction=first order; sin =0.1461)
1. 58.4 pm
2. 5.84 pm
3. 584 pm
4. 648 pm
In a compound XY, the ionic radii X+ and Y- are 88 pm and 200 pm respectively. What is the coordination number of X+?
1. 4
2. 6
3. 8
4. 10
The edge length of unit cell of a metal having molecular weight 75 g/mol is 5 Å which crystallizes in cubic lattice. If the density is 2 g/cc then find the radius of metal atom. . Give the answer in pm.
1. 116.5 pm
2. 316.5 pm
3. 216.5 pm
4. 416.5 pm
Sodium metal crystallizes as a body-centred cubic lattice with the cell edge 4.29 Å. What is the radius of sodium atom?
1.
2.
3.
4.






