the optically active tartaric acid is named as D-(+)- tartaric acid because it has a positive
1. optical rotation: and is derived from D-glucose
2. pH in an organic solvent
3. optical rotation and is derived from D-(+)-glyceraldehyde
4. optical rotation only when Substituted by deuterium
The IUPAC name of the above mentioned compound is -
1. p-Phenyl diphenyl
2. p-1-biphenyl benzene
3. 1,1',4',1''-terphenyl
4. Terphenyl
The total number of optically active isomers for are
1. 16
2. 8
3. 4
4. 2
If optical rotation produced by compound (i) is 36 then the rotation produced by compound (ii) is
(i) 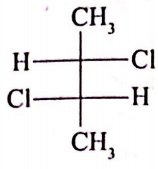
(ii) 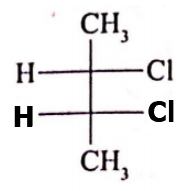
1. -36
2. 0
3. +18
4. +72
The number of the possible open chain (acyclic) isomeric compounds for molecular formula would be
1. 5
2. 6
3. 7
4. 4
The IUPAC name of the following compound is
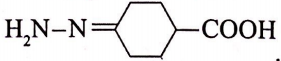
1. 4-hydrazonocyclohexane-1-carboxylic acid
2. 4-hydrazonobenzoic acid
3. 4-hydrazonocyclohexanoic acid
4. 4-(n-amino) benzene carboxylic acid
Consider the following organic compound:
To make it a chiral compound, the attack should be on carbon:
1. 1
2. 4
3. 3
4. 7
Assign the IUPAC name for the following compound.
1. 3, 4-Dichlorobenzene
2. (4-chlorophenyl)(3-chlorophenyl) diazene
3. 3,4-Bis (chlorophenyl) diazene
4. (3-Chlorophenyl)(4-chlorophenyl)diazene
Assign the IUPAC name for the following compound.
1. 3-methyl hexane -1, 2, 3-tricarbonitrile
2. hexane-1, 2, 3-tricarbonitrile
3. 1, 2-bis(cyanomethyl) butanenitrile
4. 3-(cyanomethyl)hexane-1,6-dinitrile
The number of chiral carbon atoms in the given compound is are
1. 2
2. 3
3. 4
4. 1










