Which of the following does not represent the resonating structure of 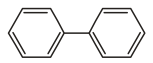
1. 
2. 
3. 
4. 


2.

3.

4.

Ordinarily the barrier to rotation about a carbon-carbon double bond is quite high but in compound P double bond between two rings was observed by NMR to have a rotational energy barrier of only about 20 cal.\mol., showing that it has lot of single bond charcter. The reason for this is
(1) Double bond having partial triple bond charcter because of resonance
(2) Doule bond undergo flipping
(3) Double bond having very high single bond charcter because of aromaticity gained in both three and five membered rings.
(4) +I effect of nC3H7 groups makes double bond having partial single bond character.
The most contributing structure in nitroethene among the following is-
1. 
2. 
3. 
4.
In which of the followig molecules all the effects namely inductive, mesomeric and hyperconjugation operate ?
(1) 


The acid strength order is:
(1) I > IV > II > III
(2) III > I > II > IV
(3) II > III > I > IV
(4) I > III > II > IV
Acid strength of the conjugate acids of the following are-
(1) I > II > III > IV
(2) III > II > I > IV
(3) IV > III > II > I
(4) None of these
The acid dissociation constants of the following acids are given as under:
Compound Ka CICH2COOH 136 × 10–5 


(i) The above variation in acidities of the above acids are due to inductive effect only.
(ii) The above variation are both due to inductive and resonance effects.
(iii) Inductive effect varies sharply with distance.
(iv) –I effect of chlorine is not much.
(1) i and ii
(2) i and iii
(3) ii and iii
(4) iii and iv
If 
1. P
2. Q
3. R
4. S
The correct acidic strength order of acidic hydrogen x, y and z is respectively.
(1) x > z > y
(2) x > y > z
(3) z > y > x
(4) y > z > x
The correct basic strength order is:
(1) I > II > IV > III
(2) IV > III > II > I
(3) III > II > IV > I
(4) III > IV > II > I













