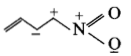
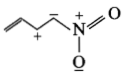
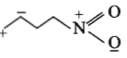
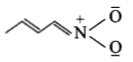
The least stable resonance structure among the following is:
1. 
2. 
3. 
4. 




Hyperconjugation involves the overlap of the following orbitals:
1. σ - σ
2. σ - p
3. p - p
4. π - π
The IUPAC name of the compound shown below is:
1. 2-bromo-6-chlorocyclohex-1-ene
2. 6-bromo-6-chlorocyclohexene
3. 3-bromo-1-chlorocyclohex-1-ene
4. 1-bromo-3-chlorocyclohexene
Which of the following structures are superimposable?
(1)
(2)
(3)
(4)
(1) 1 and 2
(2) 2 and 3
(3) 1 and 4
(4) 1 and 3
The production of an optically active compound from a symmetric molecule without resolution in termed as:
(1) Walden inversion
(2) partial racemisation
(3) asymmetric synthesis
(4) partial resolution
Buta-1,3-diene and But-2-yne are:
1. Position isomers
2. Functional isomers
3. Chain isomers
4. Tautomers
The number of different substitution products possible when ethane is allowed to react with Chlorine in sunlight are:
(1) 9
(2) 6
(3) 8
(4) 5
In butane, which of the following forms has the lowest energy?
(1) Gauche form
(2) Eclipsed form
(3) Staggered form
(4) None of these
What is the relationship between the two given molecular structures?

| 1. | Enantiomers | 2. | Position isomers |
| 3. | Conformers | 4. | None of the above |
In the following carbocations, the most stabile carbocation:
(1) RCH2C+H3
(2)
(3) 
(4)












