Maleic and fumaric acids are:
1. tautomers
2. geometrical isomers
3. chain isomers
4. functional isomers
Isomers which can be interconverted through rotation around a single bond are:
1. Conformers
2. Diastereomers
3. Enantiomers
4. Positional isomers
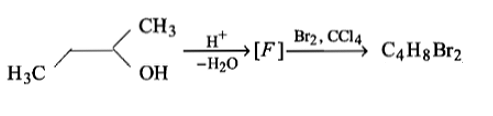
How many structures of F are possible?
(1) 2
(2) 5
(3) 6
(4) 3
The optically active alkane with lowest molar mass is:
1.
2.
3. 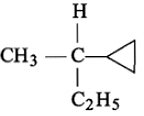
4.
a-D-(+)-glucose and β-D-(+)-glucose are :
(1) enantiomers
(2) conformers
(3) epimers
(4) anomers
The absolute configuration of

(1) S, R
(2) S, S
(3) R, R
(4) R, S
The correct statement regarding electrophile is
| 1. | Electrophile is a negatively charge species and can form a bond by accepting a pair of electrons from a nucleophile |
| 2. | Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile |
| 3. | Electrophile can be either neutral or positively charged species and can form a bond by accepting a pair of electrons from a nucleophile |
| 4. | Electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile |
Which of the following statements is true regarding the conformers of ethane?
| 1. | Bond angle remains the same but bond length changes. |
| 2. | Bond angle changes but bond length remains the same. |
| 3. | Both bond angle and bond length change. |
| 4. | Both bond angle and bond length remain the same. |
The IUPAC name of the compound

1. 3-Keto-2-methylhex-4-enal
2. 5-Formylhex-2-en-3-one
3. 5-Methyl-4-oxohex-2-en-5-al
4. 3-Keto-2-methylhex-5-enal
The correct order of acidity among the following is:
1. CH2=CH2 >CH≡CH > CH3C≡CH > CH3-CH3
2. CH≡CH > CH3-C≡CH > CH2=CH2 >CH3-CH3
3. CH≡CH > CH2=CH2 > CH3-C≡CH > CH3-CH3
4. CH3-CH3 > CH2=CH2 > CH3-C≡CH > CH≡CH






