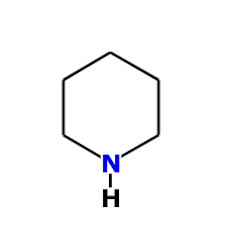
exists but does not because:
1. Nitrogen has no vacant d-subshells .
2. is unstable .
3. Nitrogen atom is much smaller than p .
4. Nitrogen is highly inert .
The bond angle between two hybrid orbitals is . The % s character in orbital is-
| 1. | Between 30-31% | 2. | Between 9- 12% |
| 3. | Between 25 -26% | 4. | Between 22-23% |
Which bond angle would result in the maximum dipole moment for the triatomic molecule
1.
2.
3.
4.
The strongest hydrogen bond present among the following is-
1.
2.
3.
4.
The ionisation energy will be maximum for the process:
(a)
(b)
(c)
(d)
In which of the process, the bond order increases and magnetic behaviour changes?
(a)
(b)
(c)
(d)
The coordination geometry around and hybridization of N and B atoms in a 1:1 complex of NH3 and BF3 :
(1) N: tetrahedral, sp3; B: tetrahedral, sp3
(2) N: pyramidal, sp3; B: pyramidal, sp3
(3) N: pyramidal, sp3; B: planar, sp2
(4) N: pyramidal, sp3; B: tetrahedral, sp3
The formation of the oxide ion (g) requries first an exothermic and then an endothermic step as shown below,
Assuming mixing is NOT operative, the paramagnetic species among the following is :
(1)
(2)
(3)
(4)