How many unit cells are present in a cube shaped ideal crystal of NaCl of mass 1.00g?
[ Atomic masses Na=23, Cl = 35.5]
1. 2.57 x 1021
2. 5.14 x 1021
3. 1.28 x 1021
4. 1.71 x 1021
The ratio of cations to anion in a closed pack tetrahedral is:
1. 0.714
2. 0.225
3. 0.02
4. none of these
In a compound, atoms of element Y form ccp lattice, and those of element X occupy 2/3rd of tetrahedral voids. The formula of the compound will be :-
1. X3Y4
2. X4Y3
3. X2Y3
4. X2Y
A spinel is an important class of oxides consisting of two types of metal ions with the oxide ions arranged in ccp layers. The normal spinel has 1/8th of the tetrahedral void occupied by one type of metal and one half of the octahedral voids occupied by another type of metal ions. Such a spinel is formed by Zn2+, Al3+ and O2- with Zn2+ in tetrahedral void. Give the simplest formula of the spinel.
1. ZnAl2O4
2. ZnAl2O3
3. ZnAlO
4. None of these
Close packing is maximum in the crystal lattice of:
1. simple cubic
2. face centred
3. body centred
4. none of these
Frenkel defect is noticed in:
1. AgBr
2. ZnS
3. Agl
4. All of the above
The density of KCl is 1.9893 g cm-3 and the length of a side unit cell is 6.29082 Å as determined by X-ray diffraction. The value of Avogadro’s number
calculated from this data is:-
1. 6.017 x 1023
2. 6.023 x 1022
3. 7.03 x 1023
4. 6.01 x 1019
How many octahedral and tetrahedral holes are present per unit cell in a face centred cubic arrangement of atoms?
1. 8,4
2. 1,2
3. 4,8
4. 2 ,1
TiO2 is a well-known example of:
1. Triclinic system.
2. Tetragonal system.
3. Monoclinic system.
4. None of the above.
For a solid with the following structure, the co-ordination number of the point B is:
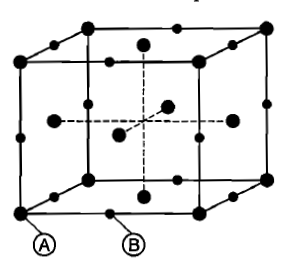
1. 3
2. 4
3. 5
4. 6






