Aniline and Ethylamine resemble in:
1. Solubility
2. Action with HNO2
3. Action of Grignard reagent
4. Coupling reaction
A secondary amine is:
1. a compound with two -NH2 groups
2. a compound with 2 carbon atoms and a -NH2 group
3. a compound with a -NH2 group on the carbon atom in number 2 position
4. a compound in which 2 of the hydrogens of NH3 have been replaced by alkyl or aryl groups

The above reaction is an example of....
1. Intermolecular C-N coupling
2. Intramolecular C-N coupling
3. Intermolecular N-N coupling
4. Intramolecular N-N coupling
Tertiary nitro compounds cannot show tautomerism because:
1. they are very stable
2. isomerises to give sec. nitro compounds
3. do not have labile H-atom
4. they are highly eactive
CH3CH2NH2 contains a basic NH2 group, but CH3CONH2 does not, because;
1. acetamide is amphoteric in character
2. in CH3CH2NH2 the electron pair on N-atom is delocalized by resonance
3. in CH3CH2NH2 there is no resonance, while in acetamide the lone pair of electron on N-atom is delocalized and therefore less available for protonation
4. none of the above
Identfy X in the sequence,
:
1.
2.
3.
4. none of the above
The end product in the following sequence of reactions is -
1. Ethyl cyanide
2. Ethylamine
3. Methylamine
4. Acetamide
Which of the following reactions does not yield an amine?
1. 
![]()
2. 

3. 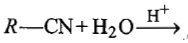
![]()
4. 

An amine reacts with and the product is soluble in alkali, amine is:
1.
2.
3.
4. all of those
Among the following, the strongest base is:
1. C6H5NH2
2. p-NO2-C6H4NH2
3. m-NO2-C2H4NH2
4. C2H5CH2NH2







