Select Chapter Topics:
Select the correct option based on statements below:
Assertion (A):
of aniline is higher than ethylamine.
Reason (R):
The lone pair of group in aniline is involved in conjugation with a benzene ring.
1.
Both (A) and (R) are true and (R) is the correct explanation of (A).
2.
Both (A) and (R) are true but (R) is not the correct explanation of (A).
3.
(A) is true but (R) is false.
4.
Both (A) and (R) are false.
Subtopic: Amines - Preparation & Properties |
85%
From NCERT
Please attempt this question first.
Hints
Links
Please attempt this question first.
Select the correct option based on statements below:
| Assertion (A): | Aniline does not undergo the Friedel-Crafts reaction. |
| Reason (R): | Diazonium salts of aromatic amines are more stable than those of aliphatic amines. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Diazonium Salts: Preparation, Properties & Uses |
70%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
Select the correct option based on statements below:
| Assertion (A): | 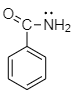 is more basic than is more basic than |
| Reason (R): | Delocalization of a lone pair of electrons decreases the basic strength. |
| 1. | Both (A) and (R) are true and (R) is not the correct explanation of (A). |
| 2. | (A) is true but (R) is false |
| 3. | .Both (A) and (R) are true but (R) is the correct explanation of (A). |
| 4. | Both (A) and (R) are false. |
Subtopic: Amines - Preparation & Properties |
55%
From NCERT
Please attempt this question first.
Hints
Links
Please attempt this question first.
Select the correct option based on statements below:
| Assertion (A): | C6H5NH2 is a 1° amine and can be prepared by Gabriel phthalimide synthesis. |
| Reason (R): | C6H5NH2 is strongly basic in nature. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Amines - Preparation & Properties |
69%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
Based on the statements given, which option is correct?
| Assertion (A): | Nitration of aniline at a low temperature gives 47% m-nitroaniline. |
| Reason (R): | In acidic medium, group is converted into group which is m-directing. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Subtopic: Mechanism |
84%
From NCERT
Please attempt this question first.
Hints
Links
Please attempt this question first.
Select the correct option based on statements below:
| Assertion (A): | Amines have a higher boiling point than the corresponding alcohols. |
| Reason (R): | Alcohols possess intramolecular H-bonding. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Amines - Preparation & Properties |
67%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
Select the correct option based on statements below:
| Assertion (A): | Secondary amines have higher boiling points than their respective tertiary isomers. |
| Reason (R): | H-bonding is absent in tertiary amines. |
| 1. | Both (A) and (R) are true and (R) is not the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Amines - Preparation & Properties |
61%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
The following reaction is the best method to introduce F to benzene:
The structure of intermediate compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Diazonium Salts: Preparation, Properties & Uses |
83%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
Whose melting point is the highest?
| 1. | Primary amines | 2. | Secondary amines |
| 3. | Tertiary amines | 4. | Quaternary amines |
Subtopic: Amines - Preparation & Properties |
69%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the following amines doesn't give the carbylamine reaction?
| 1. | Ethylamine | 2. | Dimethylamine |
| 3. | Methylamine | 4. | Phenylamine |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
80%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch



