The reagent among the following that does not reduce an aryl nitro compound to an amine is:
1. H2(excess) / Pt
2. LiAlH4 in ether
3. Fe and HCI
4. Sn and HCI
Subtopic: Urea & Nitro Compound |
61%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The reagent employed in converting nitromethane to methylamine is:
| 1. | \(\mathrm{SOCl}_2 \) | 2. | \(\mathrm{NaNH}_2 \) |
| 3. | \(\mathrm{PCl}_5 \) | 4. | \(\mathrm{Sn} / \mathrm{HCl}\) |
Subtopic: Urea & Nitro Compound |
89%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
A nitrogen-containing aromatic compound A reacts with Sn/HCl, followed by HNO2 to give an unstable compound B. On treatment with phenol, B forms a beautiful coloured compound C with the molecular formula C12H10N2O. The structure of compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Urea & Nitro Compound |
71%
From NCERT
NEET - 2016
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Consider the following reaction sequence:

The major product D in the above-mentioned reaction is:
| 1. | 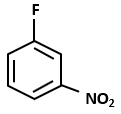 |
2. | 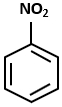 |
| 3. | 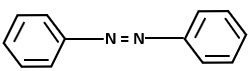 |
4. | 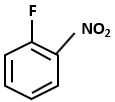 |
Subtopic: Urea & Nitro Compound |
51%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.


