Which reagent transforms nitromethane into methylamine?
1. Zn/HCl
2. Zn/NaOH
3. Zn/C2H5OH
4. Ni/H2
Subtopic: Amines - Preparation & Properties | Mechanism |
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The reagent that is used to convert acetamide into methyl amine is:
1. NaOH-Br2
2. Soda lime
3. Hot conc. H2SO4
4. PCl5
Subtopic: Amines - Preparation & Properties | Mechanism |
90%
From NCERT
AIPMT - 2010
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the following bases is the strongest in an aqueous medium?
1. CH3NH2
2. NCCH2NH2
3. (CH3)2NH
4. C6H5NHCH3
Subtopic: Mechanism |
79%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The final product C in the below mentioned reaction is:

 \(\xrightarrow[]{Ac_2O}\ A\ \xrightarrow[CH_3COOH]{Br_2}\ B\ \xrightarrow[H^+]{H_2O}\ C\)
\(\xrightarrow[]{Ac_2O}\ A\ \xrightarrow[CH_3COOH]{Br_2}\ B\ \xrightarrow[H^+]{H_2O}\ C\)
| 1. | 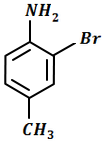 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Mechanism |
65%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the reactions given in Column-I with the statements given in Column-II.
| Column-I | Column-II | ||
| A. | Ammonolysis | (i) | Amine with a lesser number of carbon atoms |
| B. | Gabriel phthalimide synthesis | (ii) | Detection test for primary amines. |
| C. | Hofmann bromamide reaction | (iii) | Reaction of phthalimide with KOH and R—X |
| D. | Carbylamine reaction | (iv) | Reaction of alkyl halides with NH3 |
Codes:
| A | B | C | D | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (iii) | (i) | (iv) | (ii) |
| 3. | (i) | (iv) | (iii) | (ii) |
| 4. | (iv) | (iii) | (i) | (ii) |
Subtopic: Diazonium Salts: Preparation, Properties & Uses | Mechanism |
89%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The product formed from the following reaction sequence is


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Diazonium Salts: Preparation, Properties & Uses | Mechanism |
75%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Based on the statements given, which option is correct?
| Assertion (A): | Nitration of aniline at a low temperature gives 47% m-nitroaniline. |
| Reason (R): | In acidic medium, group is converted into group which is m-directing. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Subtopic: Mechanism |
85%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Benzene diazonium chloride on reaction with aniline in the presence of dilute hydrochloric acid gives:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Mechanism |
92%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch




