A pair in which both species are not likely to exist is:
1.
\(H^+_2,He^{2-}_2\)
2.
\(H^-_2,He^{2+}_2\)
3.
\(H^{2+}_2,He_2\)
4.
\(H^+_2,He^{2+}_2\)
Among the following atomic orbital overlaps, the non-bonding overlap is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Combination of atoms A and B that forms an anti-bonding molecular orbital is :
1.
2.
3.
4.
The number of (i) sp2 hybridized carbon atoms and (ii) bonds are present in the following compound are:
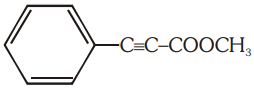
1. 7, 5
2. 8, 6
3. 7, 6
4. 8, 5
The decreasing order of ionic character of the N-H, F-H, C-H, and O-H, is :
| 1. | N-H > F-H > C-H > O-H |
| 2. | F-H > N-H > C-H > O-H |
| 3. | O-H > C-H > F-H > N-H |
| 4. | F-H > O-H > N-H > C-H |
Which of the following is the most basic oxide?
1.
2.
3.
4.
An electron-deficient compound among the following is:
| 1. | (SiH3)2 | 2. | (BH3)2 |
| 3. | PH3 | 4. | (CH3)2 |
| 1. | The H-O-H bond angle in H2O is larger than the H-C-H bond angle in CH4 |
| 2. | The H-C-H bond angle in CH4 is larger than the H-N-H bond angle in NH3 |
| 3. | The H-C-H bond angle in CH4, the H-N-H bond angle in NH3 and the H-0-H bond angle in H2O are all greater than 90o |
| 4. | The H-O-H bond angle in H2O is smaller than the H-N-H bond angle in NH3 |
The correct increasing order of ionic character of the following molecule is:
LiF, K2O, N2, SO2, ClF3
1. N2 < SO2 < ClF3 < K2O < LiF
2. N2 > SO2 > ClF3 > K2O < LiF
3. N2 > SO2 > K2O > ClF3 > LiF
4. LiF > K2O < ClF3 > SO2 > N2


