|
A |
B |
|
A. |
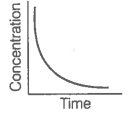
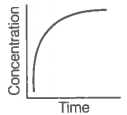
i. Variation in product concentration with time. |
|
B. |
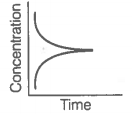
ii. Reaction at equilibrium |
|
C. |
iii. Variation in reactant concentration with time. |
Match the graphical variations with their descriptions given above and identify the correct codes below:
| A | B | C | |
| 1. | i | iii | ii |
| 2. | i | ii | iii |
| 3. | iii | ii | i |
| 4. | iii | i | ii |
A graph of volume of hydrogen released vs time for the reaction between zinc and dil. HCl is given in the graph below.

The correct statement among the following based on the graph given above is:
Consider the graph given in the figure. Which of the following options does not show an instantaneous rate of reaction in the 40s?

1. \({V_5 -V_2 \over 50-30}\)
2. \({V_4 -V_2 \over 50-30}\)
3. \({V_3 -V_2 \over 40-30}\)
4. \({V_3 -V_1 \over 40-20}\)
True statement among the following is:
| 1. | The rate of a reaction decreases with the passage of time as the concentration of reactants decreases. |
| 2. | The rate of a reaction is the same at any time during the reaction. |
| 3. | The rate of a reaction is independent of temperature change. |
| 4. | The rate of a reaction decreases with an increase in the concentration of the reactants. |
The correct expression for the rate of reaction given below is:
\(5 \mathrm{Br}^{-}(\mathrm{aq})+\mathrm{BrO}_3^{-}(\mathrm{aq})+6 \mathrm{H}^{+}(\mathrm{aq}) \rightarrow 3 \mathrm{Br}_2(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\mathrm{l})\)
| 1. | \(\frac{\Delta\left[B r^{-}\right]}{\Delta t}=5 \frac{\Delta\left[H^{+}\right]}{\Delta t} \) | 2. | \(\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta t}=\frac{6}{5} \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t} \) |
| 3. | \(\frac{\Delta[\mathrm{Br^-}]}{\Delta t}=\frac{5}{6} \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t} \) | 4. | \(\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta t}=6 \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t}\) |
Rate law for the reaction \(A+2 B \rightarrow C\) is found to be
Rate = k[A][B]
If the concentration of reactant 'B' is doubled, keeping the concentration of A constant, then the value of the rate of the reaction will be:
| 1. | The same. | 2. | Doubled. |
| 3. | Quadrupled. | 4. | Halved. |
Compounds A and B react according to the following chemical equation.
A(g) + 2B(g) 2C(g)
The concentration of either A or B was changed keeping the concentrations of one of the reactants constant and rates were measured as a function of initial concentration. The following results were obtained. Choose the correct option for the rate equations for this reaction.
| Experiment | Initial concentration of [A]/mol L–1 |
Initial concentration of [B]/moI L–1 |
Initial rate (mol L–1 s–1) |
| 1. 2. 3. |
0.30 0.30 0.60 |
0.30 0.60 0.30 |
0.10 0.40 0.20 |
2.
3.
4.
Consider the reaction AB. The concentration of both the reactant and the product varies exponentially with time.
The graph that accurately depicts how reactant and product concentrations change with time is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Rate law expression cannot be determined from a balanced chemical equation if.
| (a) | The reverse reaction is involved |
| (b) | It is an elementary reaction |
| (c) | It is a sequence of elementary reactions |
| (d) | Any of the reactants is in excess |
The correct choice among the given is -
1. (a, b, c)
2. (b, c, d)
3. (a, c, d)
4. (a, b, d)
At high pressure the following reaction is zero order-
The correct statements among the following is:
| (a) | Rate of reaction = Rate constant |
| (b) | Rate of the reaction depends on the concentration of ammonia |
| (c) | Rate of decomposition of ammonia will remain constant until ammonia disappears completely |
| (d) | Further increase in pressure will change the rate of reaction |
| 1. | (a, b, c) | 2. | (b, c, d) |
| 3. | (a, c, d) | 4. | (a, b, d) |