The half-life for radioactive decay of 14C is 5730 years. A wood sample contains only 80% of the 14C. The age of the wood sample would be-
1. 1898 years
2. 1765 years
3. 1931 years
4. 1860 years
For a reaction A → Product, with k = 2.0 × 10–2 s–1, if the initial concentration of A is 1.0 mol L–1, the concentration of A after 100 seconds would be :
| 1. | 0.23 mol L–1 | 2. | 0.18 mol L–1 |
| 3. | 0.11 mol L–1 | 4. | 0.13 mol L–1 |
The decomposition of sucrose follows the first-order rate law. For this decomposition, t1/2 is 3.00 hours. The fraction of a sample of sucrose that remains after 8 hours would be:
| 1. | 0.13 | 2. | 0.42 |
| 3. | 0.16 | 4. | 0.25 |
Given the following observations:
| Experiment | [A] / mol L–1 | [B] / mol L–1 | Initial rate / mol L–1 min–1 |
| I | 0.1 | 0.1 | 2.0 × 10–2 |
| II | X | 0.2 | 4.0 × 10–2 |
| III | 0.4 | 0.4 | Y |
The reaction between A and B is first-order with respect to A and zero-order with respect to B. The values of X and Y are, respectively:
1. X = 0.2 \(mol\) \(L^{- 1}\); Y = \(\) \(0 . 08\) \(mol\) \(L^{- 1} \left(min\right)^{- 1}\)
2. X = 0.02 \(mol\) \(L^{- 1}\); Y = \(\) \(0 . 08\) \(mol\) \(L^{- 1} \left(min\right)^{- 1}\)
3. X = 0.01 \(mol\) \(L^{- 1}\); Y = \(\) \(0 . 8\) \(mol\) \(L^{- 1} \left(min\right)^{- 1}\)
4. X = 0.2 \(mol\) \(L^{- 1}\); Y = \(\) \(0 . 8\) \(mol\) \(L^{- 1} \left(min\right)^{- 1}\)
The rate constant of a radioactive substance is . The value of half-life will be :
1. 0.05 years
2. 0.17 years
3. 0.26
4. 1.6 years
During a nuclear explosion, one of the products is 90Sr with a half-life of 28.1 years. If 1µg of 90Sr was absorbed in the bones of a newly born baby instead of calcium, the amount of 90Sr that will remain after 10 years in the now grown up child would be -
(Given ,antilog(0.108)=1.28)
1. 0.227 µg
2. 0.781 µg
3. 7.81 µg
4. 2.27 µg
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data was obtained:
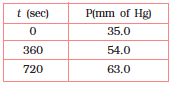
The rate constant of the above reaction would be -
| 1. | 1.21 × 10–3 s–1 | 2. | 2.21 × 10–3 s–1 |
| 3. | 3.21 × 10–3 s–1 | 4. | 4.21 × 10–3 s–1 |
For a first-order reaction, the relationship between time required for 99% completion to the time required for the completion of 90% of the reaction would be :
| 1. | 2. | ||
| 3. | 4. |
A first-order reaction takes 40 min for 30% decomposition. Half life of the reaction is-
1. 55.9 min
2. 77.9 min
3. 63.9 min
4. 80.9 min
The rate constant for a first-order reaction is . The time required to reduce the initial concentration of the reactant to its 1/16 value is-
| 1. | 2. | ||
| 3. | 4. |


