The value of equilibrium constant of the reaction HI (g) H2(g) + I2(g) is 8.0. The equilibrium constant of the reaction
H2(g) + I2(g) 2HI(g) will be
Which one of the following ionic species has the greatest proton affinity to form stable compound?
(1) HS-
(2)
(3) F-
(4) I-
For the reaction,
CH4(g) + 2O2(g) CO2(g) + 2H2O (l),
rH = -170.kJ mol-1
Which of the following statements is not true?
(a) At equilibrium, the concentrations of CO2(g) and H2O(l) are not equal
(b) The equilibrium constant for the reaction is given by Kp = [CO2]/[CH4][O2]
(c) Addition of CH4(g) or O2 (g) at equilibrium will cause a shift to the right
(d) The reaction is exothermic
The hydrogen ion concentration of a 10-8 M HCl aqueous solution at 298 K (Kw = 10-14) is:
(1) 1.0×10-6 M
(2) 1.0525 x 10-7 M
(3) 9.525 x 10-8 M
(4) 1.0×10-8 M
The equilibrium constant for the reaction
![]() is given by
is given by
(1)
(2)
(3)
(4)
Two flasks A and B of equal volume containing 1 mole and 2 mole of O3 respectively, are heated to the sametemperature. When the reaction 2O3 3O2 practically stops, then both the flasks shall have
(1) the same ratio [O2] /[O3]
(2) the same ratio : [O2]3/2/[O3]
(3) only O2
(4) the same time to reach equilibrium
A 10L container at 300K contains CO2 gas at pressure of 0.2 atm and an excess solid CaO (neglect thevolume of solid CaO). The volume of container is now decreased by moving the movable piston fitted in the container. What will be the maximum volume of container when pressure of CO2 attains its maximum value given that CaCO3 (s) CaO(s) + CO2(g)Kph = 0.800 atm
1. 5 L
2. 2.5 L
3. 1 L
4. The information is insufficient.
An equilibrium mixture at 700 K of 0.50 M N2, 3.00 M H2 and 2.00 M NH3 is present in a container. Now if this equilibrium is disturbed by adding N2 so that its concentration becomes 1.50 M just after addition then which of the following graphs represents the above situation more appropriately –
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
(1) 
(2)
(3)
(4) 
At 1400 K, Kc= 2.5x10-3 for the reaction CH4(g) + 2H2S(g) CS2(g) + 4H2(g). A 10.0L reaction vesselat 1400 K contains 2.00 mole of CH4, 3.0 mol of CS2, 3.0 mole of H2 and 4.0 mole of H2S. Then
| 1. | This reaction, is at equilibrium with above concentrations. |
| 2. | The reaction will proceed in forward direction to reach equilibrium. |
| 3. | The reaction will proceed in backward direction to reach equilibrium. |
| 4. | The information is insufficient to decide the direction of progress of reaction. |
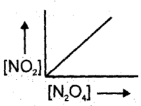
The graph which will be representing all the equilibrium concentrations for the reaction N2O4(g) 2NO2 (g) will be : (the concentrations of N2O4 (g) and of NO (g) for which the following reaction will be at equilibrium will lie:
(1) 
(2) 
(3) 
(4) 








