A compound with the molecular formula C5H10 that yields acetone on ozonolysis is:
1. 2-Methyl-2-butene
2. 2-Methyl-1-butene
3. Cyclopentane
4. 3-Methyl-1-butene
1. 2-Methyl-2-butene
2. 2-Methyl-1-butene
3. Cyclopentane
4. 3-Methyl-1-butene
A carbonyl compound reacts with hydrogen cyanide to form cyanohydrins, which form a racemic mixture of α-hydroxy acid on hydrolysis. The carbonyl compound is:
1. Acetaldehyde
2. Acetone
3. Diethyl ketone
4. Formaldehyde
The nucleophilic addition reaction will be most favored among the given compounds is:
1.
2.
3.
4.
The self-condensation product of two moles of ethyl acetate in the presence of sodium ethoxide yield is:
1. Ethyl butyrate
2. Acetoacetic ester
3. Methyl acetoacetate
4. Ethyl propionate
The product of the below-mentioned reaction is:
| 1. | 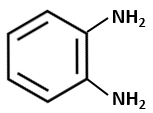 |
2. |  |
| 3. |  |
4. |  |
Reduction of aldehydes and ketones into hydrocarbons using amalgam and conc. HCl is called:
1. Clemmensen reduction
2. Cope reduction
3. Dow reduction
4. Wolff-Kishner reduction
1. Alkene
2. Amine
3. Ketone
4. Alkane
In a basic medium, acetophenone yields a stable compound A with C2H5ONa.
The structure of A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The compound that is most susceptible to a nucleophilic attack in the carbonyl group is:
1.
2.
3.
4.
The example of a nucleophilic substitution reaction among the following is:
| 1. | |
| 2. |  |
| 3. | |
| 4. |  |







